Nerve growth factor (NGF), the founder of the neurotrophin superfamily, exerts a wide range of physiological functions not only in the nervous system but also in non-neuronal cells2. It promotes neuronal cell survival or cell death, depending on the context of the neuronal environment, by interacting with two structurally unrelated classes of receptors: tropomyosine receptor kinase A (TrkA), which is specific for NGF, and/or p75NTR, which is common to all the neurotrophins8. Apart from being involved in both the development and the maintenance of specific peripheral and central neuronal populations, NGF is also implicated in several disease states, such as certain chronic inflammatory and neuropathic pain states and several human malignancies11. Moreover, NGF has been shown to be an autocrine factor essential for the survival of macrophages infected with HIV1. Therefore, the involvement of NGF in many pathological states unrelated with the central nervous system raised the possibility that NGF antagonists could have a therapeutic effect.
Several preclinical studies have shown that blocking NGF can be efficacious in attenuating several types of malignant and non-malignant skeletal pain. Anti-NGF sequestering/neutralizing antibodies have been shown to reduce bone cancer pain due to sarcoma13, breast3 and prostate cancer4 as well as in the painful arthritic knee joint14, osteoarthritis10, femoral fracture5, and low back pain6.
Sabsovich, I. et al.12 investigated the effect of Anti-NGF Antibody treatment in rat model of complex regional pain syndrome type I (CRPS I). This model exhibits the classical signs of CRPS type I including allodynia, edema, warmth, and periarticular bone loss. It was found that Anti-NGF Antibody treatment reduced neuropeptide levels in sciatic nerve and reduced nociceptive sensitization.
A recent study demonstrated that blocking the activity of NGF in the injured spinal cord with a neutralizing antibody greatly reduced the autonomic dysreflexia7. Autonomic dysreflexia characterized by a potentially life-threatening increase in arterial pressure that can cause debilitating headaches, seizures, strokes, and even death9. Neutralizing NGF in the spinal cord is a promising strategy to minimize the life-threatening autonomic dysreflexia that develops after spinal cord injury.
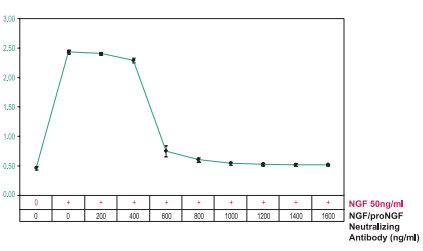
PC12 cells were grown in the absence of serum and in the presence of Native mouse NGF 2.5S protein (99%) (#N-240), (50 ng/ml) as indicated. Varying concentrations of Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) were added to the culture and cell viability was established after 24 h using the XTT method. Note that 600 ng/ml Mouse NGF/proNGF Neutralizing Antibody (#ALM-006), was able to completely block the activity of 50 ng/ml mouse NGF.
NGF neutralization is without a doubt an important therapeutic goal in various pathological states. Alomone Labs is pleased to offer a mouse monoclonal antibody with NGF/proNGF neutralizing activities. Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) is directed against an epitope of mouse NGF. Mouse NGF/proNGF Neutralizing Antibody was shown to block the biological effects of NGF and proNGF in ELISA assays. The antibody was designed to recognize NGF and proNGF from human, mouse and rat samples.
References
- Balestra, E. et al. (2001) J. Biol. Regul. Homeost. Agents 15, 272.
- Bibel, M. and Barde, Y.A. (2000) Genes Dev. 14, 2919.
- Bloom, A.P. et al. (2011) J. Pain 12, 698.
- Halvorson, K.G. et al. (2005) Cancer Res. 65, 9426.
- Jimenez-Andrade, J.M. et al. (2007) Pain 133, 183.
- Katz, N. et al. (2011) Pain 152, 2248.
- Krenz, N.R. et al. (1999) J. Neurosci. 19, 7405.
- Lewin, G.R. and Barde, Y.A. (1996) Annu. Rev. Neurosci. 19, 289.
- Mathias, C.J., The cardiovascular system in tetraplegia and paraplegia., in Handbook of clinical neurology. 1992, Elsevier Science. p. 435.
- McNamee, K.E. et al. (2010) Pain. 149, 386.
- Pezet, S. and McMahon, S.B. (2006) Annu. Rev. Neurosci. 29, 507.
- Sabsovich, I. et al. (2008) Pain 138, 47.
- Sevcik, M.A. et al. (2005) Pain 115, 128.
- Shelton, D.L. et al. (2005) Pain 116, 8.