Overview
- Peptide (C)RHLGTIPRLSLSR, corresponding to amino acid residues 27-39 of rat Glial fibrillary acidic protein (Accession P47819). Intracellular, cytoplasm.

 Western blot analysis of rat brain membranes (lanes 1 and 3), (1:2000) and mouse brain lysate (lanes 2 and 4), (1:400):1,2. Anti-GFAP Antibody (#AFP-001).
Western blot analysis of rat brain membranes (lanes 1 and 3), (1:2000) and mouse brain lysate (lanes 2 and 4), (1:400):1,2. Anti-GFAP Antibody (#AFP-001).
3,4. Anti-GFAP Antibody, preincubated with GFAP Blocking Peptide (#BLP-FP001).
 Expression of GFAP in rat brainImmunohistochemical staining of free-floating rat frozen brain sections using Anti-GFAP Antibody (#AFP-001), (1:2000). In rat parietal cortex, GFAP immunoreactivity (green) appears in astrocytes (arrows). Nuclei are stained using DAPI as the counterstain (blue).
Expression of GFAP in rat brainImmunohistochemical staining of free-floating rat frozen brain sections using Anti-GFAP Antibody (#AFP-001), (1:2000). In rat parietal cortex, GFAP immunoreactivity (green) appears in astrocytes (arrows). Nuclei are stained using DAPI as the counterstain (blue).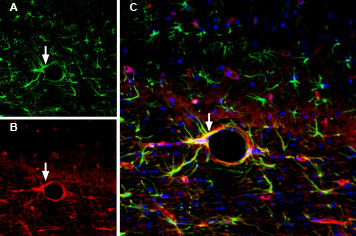 Multiplex staining of GFAP and AQP4 in rat brainImmunohistochemical staining of immersion-fixed, free floating rat brain frozen sections using Anti-GFAP Antibody (#AFP-001), (1:2000) and Anti-Aquaporin 4 (AQP4) (300-314)-ATTO Fluor-594 Antibody (#AQP-014-AR), (1:100). A. In rat fornix, GFAP immunoreactivity (green) appears in astrocytic processes. B. In same section, AQP4 staining (red) is detected in blood vessels. C. Merge of panel A and panel B shows colocalization around a large blood vessel (arrows). Nuclei are stained using DAPI as the counterstain (blue).
Multiplex staining of GFAP and AQP4 in rat brainImmunohistochemical staining of immersion-fixed, free floating rat brain frozen sections using Anti-GFAP Antibody (#AFP-001), (1:2000) and Anti-Aquaporin 4 (AQP4) (300-314)-ATTO Fluor-594 Antibody (#AQP-014-AR), (1:100). A. In rat fornix, GFAP immunoreactivity (green) appears in astrocytic processes. B. In same section, AQP4 staining (red) is detected in blood vessels. C. Merge of panel A and panel B shows colocalization around a large blood vessel (arrows). Nuclei are stained using DAPI as the counterstain (blue). Multiplex staining of Connexin-43 and GFAP in rat cerebellum.Immunohistochemical staining of perfusion-fixed frozen rat brain sections with Guinea pig Anti-Connexin-43 Antibody (#ACC-201-GP), (1:200), followed by goat anti-guinea pig-AlexaFluor-594 and Anti-GFAP Antibody (#AFP-001), (1:1200), followed by goat anti-rabbit-AlexaFluor-488. A. Connexin-43 immunoreactivity (red) appears as positive puncta (arrows). B. GFAP immunoreactivity (green) appears along the Bergmann glial processes (arrows point at examples). C. Merge of the two images shows CNX-43 puncta distributed along the Bergmann glial processes. Cell nuclei are stained with DAPI (blue).
Multiplex staining of Connexin-43 and GFAP in rat cerebellum.Immunohistochemical staining of perfusion-fixed frozen rat brain sections with Guinea pig Anti-Connexin-43 Antibody (#ACC-201-GP), (1:200), followed by goat anti-guinea pig-AlexaFluor-594 and Anti-GFAP Antibody (#AFP-001), (1:1200), followed by goat anti-rabbit-AlexaFluor-488. A. Connexin-43 immunoreactivity (red) appears as positive puncta (arrows). B. GFAP immunoreactivity (green) appears along the Bergmann glial processes (arrows point at examples). C. Merge of the two images shows CNX-43 puncta distributed along the Bergmann glial processes. Cell nuclei are stained with DAPI (blue).
- Eng, L.F. et al. (2000) Neurochem. Res. 25, 1439.
- Herrmann H and Aebi, U. (1998) Curr. Opin. Struct. Biol. 8, 177.
- Yang, Z. et al. (2015) Trends Neurosci. 38, 364.
- Prust, M. et al. (2011) Neurology 77, 1287.
- Kamphuis, W. et al. (2014) Neurobiol. Aging 35, 492.
Glial fibrillary acidic protein (GFAP) is a key intermediate filament (IF) III protein responsible for maintaining the mechanical strength of glia cells by supporting their cytoskeleton structure. GFAP is expressed in astrocytes in the CNS, non-myelinating Schwann cells in the PNS, and enteric glial cells1.
GFAP has a structural organization that is typical to class III IF proteins: it has a head, rod, and tail domains. The N-terminal head domain is important for filament formation and the C-terminal domain is important for oligomerization2.
GFAP is encoded by a single gene mapped to human chromosome 17q21. To date, 10 isoforms/splice variants have been identified. GFAP is tightly regulated: both at mRNA transcription level and by phosphorylation and other post-translational modifications. A number of growth factors such as CNTF, FGF and TGF-β can induce GFAP gene transcription activation leading to increased GFAP protein levels3.
Single nucleotide polymorphism (SNP) in GFAP results in the formation of Rosenthal fibers that cause Alexander Disease, hence, GFAP is a potential drug target for the treatment of this disease. A number of GFAP mutations were found in the coding and in the promoter regions of Alexander disease patients4.
GFAP gene activation and protein induction appear to play a critical role in astroglia cell activation (astrogliosis) following CNS injuries and neurodegeneration. GFAP protein and its breakdown products are rapidly released into biofluids following traumatic brain and spinal cord injuries and stroke, making them strong candidate biomarkers for such neurological disorders5.
Application key:
Species reactivity key:
Anti-GFAP Antibody (#AFP-001) is a highly specific antibody directed against an epitope of the rat Glial fibrillary acidic protein. The antibody can be used in western blot and immunohistochemistry applications. It has been designed to recognize GFAP from rat and mouse samples. The antibody will not recognize human GFAP.
