Overview
- GST fusion protein with the sequence HGHLHDSAEERRLIAEGDASPG EDRTPPGLAAEPERP, corresponding to amino acid residues 119-155 of human HCN4 (Accession Q9Y3Q4). Intracellular, N-terminus.
- Rat brain membranes (1:200). Human HCN4 transfected in HEK-293T cells. (Nof, E. et al. (2007) Circulation 116, 463.).
 Western blot analysis of rat brain membranes:1. Anti-HCN4 Antibody (#APC-052), (1:200).
Western blot analysis of rat brain membranes:1. Anti-HCN4 Antibody (#APC-052), (1:200).
2. Anti-HCN4 Antibody, preincubated with HCN4 Blocking Peptide (#BLP-PC052).
- Rat thalamus lysates (Whitaker, G.M. et al. (2007) J. Biol. Chem. 282, 22900.).
- Mouse brain sections.
- HEK-293 transfected cells (1:200) (see Altomare, C. et al. (2003) J. Physiol. 549, 347.).
- Mouse sinoatrial myocytes (1:200) (St. Clair, J.R. et al. (2015) Am. J. Physiol. 309, H490).
Hyperpolarization-activated cation currents (Ih) appear in the heart and the brain and have a crucial role in controlling electrical pacemaker activity, contributing to biological processes such as heartbeat, sleep-wake cycle and synaptic plasticity.1,2
The Ih currents are generated by the Hyperpolarization-activated cyclic nucleotide-gated channel family (HCN), which is comprised of four homologous members, HCN1-4.
Each HCN subunit consists of six transmembrane domains (TM), a pore region between TM5-TM6 and a binding domain for cyclic nucleotides (CNBD) in the cytoplasmic C-terminus.2
The HCN subunits can form functional homomers and can also co-assemble into functional heteromers.2
The channels are closely related to each other and share a homology of about 60%. However, their similarity decreases in the cytoplasmic N- and C-termini. The HCN1-4 channels mainly differ from each other in their speed of activation and the extent to which they are modulated by cAMP. HCN1, weakly affected by cAMP, is the fastest channel, followed by HCN2, HCN3 and HCN4.
HCN4 is highly expressed in a restricted manner in adult sinoatrial (SA) node, constituting a good molecular marker for the adult cardiac pacemaker and might serve as a unique marker of the developing SA node .4,5
mRNA expression of HCN4 is most abundant in medial habenula and anterior and principal relay nuclei of the thalamus.6
Application key:
Species reactivity key:
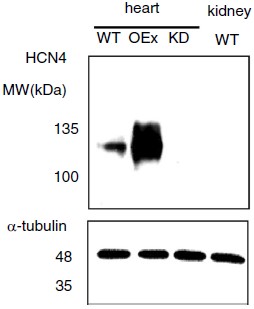
Knockout validation of Anti-HCN4 Antibody in mouse heart.Western blot analysis of mouse heart lysate using Anti-HCN4 Antibody (#APC-052). HCN4 immunodetection in wild-type, overexpressed, and knockdown animals. HCN4 expression is abolished in knockdown animals. In addition, kidney lysates were loaded as a negative control, a tissue which does not express HCN4. α-tubulin is a loading control.Adapted from Kozasa, Y. et al. (2018) J. Physiol. 596, 809. with permission of The Physiological Society.
