Overview
- Peptide (C)KNDKSDPKVRVKDD, corresponding to amino acid residues 85-98 of rat β4 subunit of voltage-gated Na+ channels (Accession Q7M730). Extracellular, N-terminus.
- Rat brain, rat cortex, mouse brain and human brain neuroblastoma cell (SH-SY5Y) lysates (1:800-1:2000).
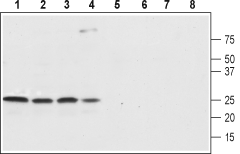 Western blot analysis of rat brain (lanes 1 and 5), rat cortex (lane 2 and 6), mouse brain (lanes 3 and 7) and SH-SY5Y (lanes 4 and 8) lysates:1-4. Anti-SCN4B (NaVβ4) (extracellular) Antibody (#ASC-044), (1:800).
Western blot analysis of rat brain (lanes 1 and 5), rat cortex (lane 2 and 6), mouse brain (lanes 3 and 7) and SH-SY5Y (lanes 4 and 8) lysates:1-4. Anti-SCN4B (NaVβ4) (extracellular) Antibody (#ASC-044), (1:800).
5-8. Anti-SCN4B (NaVβ4) (extracellular) Antibody, preincubated with SCN4B/Navβ4 (extracellular) Blocking Peptide (#BLP-SC044).
- Rat dorsal root ganglia.
- Rat living PC12 cells (1:60).
Voltage-gated sodium channels (NaV) are essential for the generation of action potentials and for cell excitability1. NaV channels are activated in response to depolarization and selectively allow the flow of Na+ ions. To date, nine NaV α subunits have been cloned and named NaV1.1-NaV1.94,5. NaV channels are classified into two groups according to their sensitivity to tetrodotoxin (TTX): TTX-sensitive (NaV1.1, NaV1.2, NaV1.3, NaV1.4, NaV1.6 and NaV1.7) and TTX-resistant (NaV1.5, NaV1.8 and NaV1.9) 2,3.
Mammalian Na+ channels are heterotrimers, composed of a central, pore-forming α subunit and two auxiliary β subunits. The expression of the α subunit isoform is developmentally regulated and tissue specific. Na+ channels in the adult central nervous system and heart contain β1 through β4 subunits, whereas Na+ channels in adult skeletal muscle have only the β1 subunit6,7.
NaVβ4 has been associated with Long QT syndrome8 and with Huntington's disease, showing a distinct down regulation of NaVβ4 in the presymptomatic stage of HD mice, whereas other voltage-gated ion channel subunits were later decreased9.

