Overview
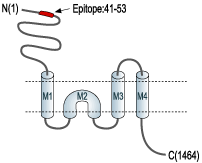
- Peptide GHSHDVTERELRN(C), corresponding to amino acid residues 41-53 of rat NR2A (Accession Q00959). Extracellular, N-terminus.
 Western blot analysis of rat brain lysates:1. Anti-NMDAR2A (GluN2A) (extracellular) Antibody (#AGC-002), (1:600).
Western blot analysis of rat brain lysates:1. Anti-NMDAR2A (GluN2A) (extracellular) Antibody (#AGC-002), (1:600).
2. Anti-NMDAR2A (GluN2A) (extracellular) Antibody, preincubated with NMDAR2A/GluN2A (extracellular) Blocking Peptide (#BLP-GC002).
 Immunoprecipitation of rat brain lysates1. Cell lysate.
Immunoprecipitation of rat brain lysates1. Cell lysate.
2. Cell lysates + protein A beads + Anti-NMDAR2A (GluN2A) (extracellular) Antibody (#AGC-002).
3. Cell lysates + protein A beads + pre-immune rabbit serum.
Black arrow indicates the NR2A protein while the red arrow shows the IgG heavy chain. Immunoblot was performed with Anti-NMDAR2A (GluN2A) (extracellular) Antibody.
 Multiplex staining of GluN2A and GluN2B in mouse deep cerebellar nucleusImmunohistochemical staining of perfusion-fixed frozen mouse brain sections using Anti-NMDAR2B (GluN2B) (extracellular)-ATTO Fluor-594 Antibody (#AGC-003-AR), (1:60) and Anti-NMDAR2A (GluN2A) (extracellular) Antibody (#AGC-002), (1:200). A. Sections were incubated with Anti-NMDAR2A (GluN2A) (extracellular) Antibody, followed by goat anti-rabbit-Alexa-488 (green). B. The same sections were incubated with Anti-NMDAR2B (GluN2B) (extracellular)-ATTO Fluor-594 Antibody (red). C. Merge of A and B demonstrates the ubiquitous colocalization of the GluN2A and GluN2B subunits in cells with neuronal profiles in this nucleus. Arrows point at an example of NR2A and NR2B co-expression.
Multiplex staining of GluN2A and GluN2B in mouse deep cerebellar nucleusImmunohistochemical staining of perfusion-fixed frozen mouse brain sections using Anti-NMDAR2B (GluN2B) (extracellular)-ATTO Fluor-594 Antibody (#AGC-003-AR), (1:60) and Anti-NMDAR2A (GluN2A) (extracellular) Antibody (#AGC-002), (1:200). A. Sections were incubated with Anti-NMDAR2A (GluN2A) (extracellular) Antibody, followed by goat anti-rabbit-Alexa-488 (green). B. The same sections were incubated with Anti-NMDAR2B (GluN2B) (extracellular)-ATTO Fluor-594 Antibody (red). C. Merge of A and B demonstrates the ubiquitous colocalization of the GluN2A and GluN2B subunits in cells with neuronal profiles in this nucleus. Arrows point at an example of NR2A and NR2B co-expression. Expression of NR2A in rat hippocampusImmunohistochemical staining of rat hippocampal dentate gyrus with Anti-NMDAR2A (GluN2A) (extracellular) Antibody (#AGC-002). A. NMDAR2A (green) appears diffusely in the outer molecular layer of the dentate gyrus (Out Mol.) and in cells along the subgranular layer (arrows). B. Staining of parvalbumin (PV, red) identifies interneurons in the dentate gyrus. C. Confocal merge demonstrates localization of PV in some neurons with NMDAR2A.
Expression of NR2A in rat hippocampusImmunohistochemical staining of rat hippocampal dentate gyrus with Anti-NMDAR2A (GluN2A) (extracellular) Antibody (#AGC-002). A. NMDAR2A (green) appears diffusely in the outer molecular layer of the dentate gyrus (Out Mol.) and in cells along the subgranular layer (arrows). B. Staining of parvalbumin (PV, red) identifies interneurons in the dentate gyrus. C. Confocal merge demonstrates localization of PV in some neurons with NMDAR2A.
 Expression of NR2A in rat C6 glioma cellsCell surface detection of NR2A in live intact rat C6 glioma cells. A. Cells were stained with Anti-NMDAR2A (GluN2A) (extracellular) Antibody (#AGC-002), (1:100), followed by goat anti-rabbit-AlexaFluor-555 secondary antibody (red). Cell nuclei were stained with the cell permeable dye Hoechst 33342 (blue staining). B. Live view of the same field.
Expression of NR2A in rat C6 glioma cellsCell surface detection of NR2A in live intact rat C6 glioma cells. A. Cells were stained with Anti-NMDAR2A (GluN2A) (extracellular) Antibody (#AGC-002), (1:100), followed by goat anti-rabbit-AlexaFluor-555 secondary antibody (red). Cell nuclei were stained with the cell permeable dye Hoechst 33342 (blue staining). B. Live view of the same field.
- Dingledine, R. et al. (1999) Pharmacol. Rev. 51, 7.
- Mayer, M.L. and Armstrong, N. (2004) Annu. Rev. Physiol. 66, 161.
- Prybylowski, K. and Wenthold, R.J. (2004) J. Biol. Chem. 279, 9673.
- Mayer, M.L. (2006) Nature 440, 456.
The NMDA receptors are members of the glutamate receptor family of ion channels that also include the AMPA and Kainate receptors.
The NMDA receptors are encoded by seven genes: one NMDAR1 (or NR1) subunit, four NR2 (NR2A-NR2D) and two NR3 (NR3A-NR3B) subunits. The functional NMDA receptor appears to be a heterotetramer composed of two NMDAR1 and two NMDAR2 subunits. Whereas the NMDAR2 subunits that assemble with the NMDAR1 subunit can be either of the same kind (i.e. two NMDAR2A subunits) or different (one NMDAR2A with one NMDAR2B). NMDAR3 subunits can substitute the NMDAR2 subunits in their complex with the NMDAR1 subunit.
The NMDAR is unique among ligand-gated ion channels in that it requires the simultaneous binding of two obligatory agonists: glycine and glutamate that bind to the NMDAR1 and NMDAR2 binding sites respectively. Another unique characteristic of the NMDA receptors is their dependence on membrane potential. At resting membrane potentials the channels are blocked by extracellular Mg2+. Neuronal depolarization relieves the Mg2+ blockage and allows ion influx into the cells. NMDA receptors are strongly selective for Ca2+ influx differing from the other glutamate receptor ion channels that are non-selective cation channels.
Ca2+ entry through the NMDAR regulates numerous downstream signaling pathways including long term potentiation (a molecular model of memory) and synaptic plasticity that may underlie learning. In addition, the NMDA receptors have been implicated in a variety of neurological disorders including epilepsy, ischemic brain damage, Parkinson’s and Alzheimer’s disease.
NMDA receptors expression and function are modulated by a variety of factors including receptor trafficking to the synapses and internalization as well as phosphorylation and interaction with other intracellular proteins.
Application key:
Species reactivity key:
Anti-NMDAR2A (GluN2A) (extracellular) Antibody (#AGC-002) is a highly specific antibody directed against an epitope of the rat protein. The antibody can be used in western blot, immunoprecipitation, immunocytochemistry, and immunohistochemistry. The antibody recognizes an extracellular epitope and is thus ideal for detecting GluN2A in living cells. It has been designed to recognize GluN2A from rat, mouse, and human samples.
Expression of NR2A in human-induced pluripotent stem cell-derived neurons.Immunocytochemical staining of human-induced pluripotent stem cell-derived neurons using Anti-NMDAR2A (GluN2A) (extracellular) Antibody (#AGC-002). Adapted from Telezhkin, V. et al. (2016) Am. J. Physiol. 310, C250. with permission of The American Physiological Society.
Applications
Citations
- Mouse brain lysate (1:2000).
Konstantoudaki, X. et al. (2016) Neuroscience 322, 333. - Mouse hippocampus lysate (1:1000).
Tindi, J.O. et al. (2015) J. Neurosci. 35, 8986. - Mouse brain lysate (1:500).
Atkin, G. et al. (2015) J. Neurosci. 35, 6165.
- Rat hippocampal neurons.
Dupuis, J.P. et al. (2014) EMBO J. 33, 842. - Rat hippocampal primary neurons (1:500).
Mikasova, L. et al. (2012) Brain 135, 1606.
- Mouse brain sections (1:100).
Atkin, G. et al. (2015) J. Neurosci. 35, 6165.
- Mouse primary astroglial cultures.
Rocchio, F. et al. (2019) Cell Death Dis. 10, 24. - HEK293 transfected cells.
Sibarov, D.A. et al. (2017) Front. Cell. Neurosci. 11, 155. - Human induced pluripotent stem cells (1:300).
Telezhkin, V. et al. (2016) Am. J. Physiol. 310, C250. - Mouse hippocampal neurons.
Atkin, G. et al. (2015) J. Neurosci. 35, 6165. - Rat cultured hippocampal neurons (1:100).
Swanger, S.A. et al. (2013) J. Neurosci. 33, 8898.
- Papouin, T. et al. (2012) Cell 150, 633.

