Overview
- Peptide (C)HLRGQRWPFGEAA(S)R, corresponding to amino acid residues 136-150 of human PAR-4 (Accession Q96RI0). Cys 149 was replaced with Ser. 1st extracellular loop.

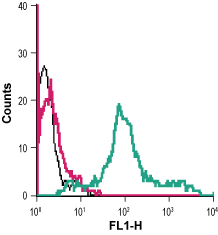 Cell surface detection of PAR-4 in live intact human MEG-01 megakaryoblastic leukemia cells:___ Cells alone.
Cell surface detection of PAR-4 in live intact human MEG-01 megakaryoblastic leukemia cells:___ Cells alone.
___ Cells + rabbit IgG isotype control-FITC.
___ Cells + Anti-PAR4 (F2RL3) (extracellular)-FITC Antibody (#APR-034-F), 5µg. Cell surface detection of PAR-4 in live intact HL-60 (human promyelocytic leukemia) cell line:___ Unstained cells.
Cell surface detection of PAR-4 in live intact HL-60 (human promyelocytic leukemia) cell line:___ Unstained cells.
___ Cells + Anti-PAR4 (F2RL3) (extracellular)-FITC Antibody (#APR-034-F).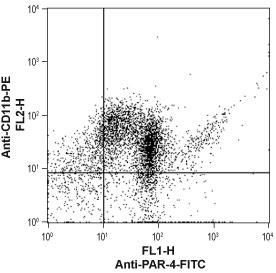 Cell surface detection of PAR-4 in human neutrophil-like differentiated promyelocytic leukemia HL-60 cellsHL-60 cells were induced to differentiate into a mature neutrophil-like phenotype by incubation with dimethyl sulfoxide (DMSO) (1.25%) for three days. The neutrophil phenotype was confirmed by staining the cells with anti-CD11b-PE antibody. Double staining with Anti-PAR4 (F2RL3) (extracellular)-FITC Antibody (#APR-034-F), (1:40), shows that most differentiated cells express both markers (upper right panel).
Cell surface detection of PAR-4 in human neutrophil-like differentiated promyelocytic leukemia HL-60 cellsHL-60 cells were induced to differentiate into a mature neutrophil-like phenotype by incubation with dimethyl sulfoxide (DMSO) (1.25%) for three days. The neutrophil phenotype was confirmed by staining the cells with anti-CD11b-PE antibody. Double staining with Anti-PAR4 (F2RL3) (extracellular)-FITC Antibody (#APR-034-F), (1:40), shows that most differentiated cells express both markers (upper right panel).- The control antigen is not suitable for this application.
- MacFarlane, S.R. et al. (2001) Pharmacol. Rev. 53, 245.
- Hollenberg, M.D. et al. (2002) Pharmacol. Rev. 54, 203.
- Ossovskaya, V.S. et al. (2004) Physiol. Rev. 84, 579.
Protease-activated receptor 4 (PAR-4) belongs to a family of four G protein-coupled receptors (PAR1-4) that are activated as a result of proteolytic cleavage by certain serine proteases, hence their name. In this novel modality of activation, a specific protease cleaves the PAR receptor within a defined sequence in its extracellular N-terminal domain. This results in the creation of a new N-terminal tethered ligand, which subsequently binds to a site in the second extracellular loop of the same receptor. This binding results in the coupling of the receptor to G proteins and in the activation of several signal transduction pathways.1-3
Different PARs are activated by different proteases. Hence, PAR-4 is activated by both thrombin and trypsin whereas PAR-1 and PAR-3 are activated only by thrombin and PAR-2 is activated only by trypsin.1-3 PAR-4 can be also cleaved and activated by other proteases such as cathepsin G.
The intracellular signaling mechanisms mediated by PAR-4 activation are not completely elucidated but they involve calcium mobilization downstream of phospholipase Cβ through the Gαq pathway.1-3
Tissue distribution of PAR-4 is very broad with the highest expression levels found in lung, testis, pancreas and small intestine. In addition, PAR-4 expression was observed in platelets, megakaryocytes and leukocytes. Studies with platelets derived form PAR-4 knockout mice have established an essential role for PAR-4 in thrombin-induced platelet activation.
PAR-4 is likely involved in other physiological functions such as regulation of gastrointestinal motility and regulation of vascular endothelial cell function.1-3
Application key:
Species reactivity key:
Anti-PAR4 (F2RL3) (extracellular) Antibody (#APR-034) is a highly specific antibody directed against an extracellular epitope of human protease-activated receptor-4. The antibody can be used in western blot analysis, immunohistochemical, immunocytochemical and flow cytometry applications. It has been designed to recognized PAR-4 from human, mouse and rat samples.
Anti-PAR4 (F2RL3) (extracellular)-FITC Antibody (#APR-034-F) is directly conjugated to fluorescein isothiocyanate (FITC). This labeled antibody can be used in immunofluorescent applications such as direct live cell flow cytometry.
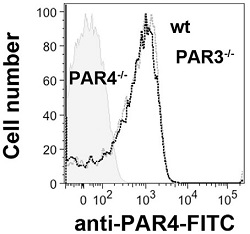
Expression of PAR-4 on mouse platelets.Direct flow cytometry analysis of PAR-4 expression in wild type (black lines), PAR3-/- (grey line), and PAR4-/- (shaded) mice platelets using Anti-PAR4 (F2RL3) (extracellular)-FITC Antibody (#APR-034-F). Note the lack of PAR-4 detection in PAR4-/- mice.Adapted from Arachiche, A. et al. (2013) PLoS ONE 8, e55740. with permission of PLoS.
Applications
Citations
- Direct flow cytometry analysis of mouse platelets. Tested in PAR4-/- mice platelets.
Arachiche, A. et al. (2013) PLoS ONE 8, e55740.
- Human macrophage U937 cells.
Mahajan-Thakur, S. et al. (2014) J. Leukoc. Biol. 96, 611. - Mouse platelets. Also tested in PAR4-/- mice platelets.
Arachiche, A. et al. (2013) PLoS ONE 8, e55740. - Human peripheral blood mononuclear cells (PBMCs).
Nieuwenhuizen, L. et al. (2013) Scand. J. Immunol. 77, 339.
