α-Bungarotoxin is a 74 amino acid peptidyl toxin isolated from the venom of the banded krait snake, Bungarus multicinctus1.
α-Bungarotoxin blocks postsynaptic neuromuscular transmission via competitive inhibition of nicotinic ACh receptors (nAChRs) with an IC50 of 3.5 x 10-10 M, thereby prevents the depolarizing action on postsynaptic membranes and blocks neuromuscular transmission2.
The toxin is selective for nAChR α7 receptors (IC50 value of 1.6 nM) and nAChR α3/β4 receptors (IC50 value of > 3 μM)3,4. α-Bungarotoxin also binds to and blocks a subset of GABAA receptors (GABAARs) that contain the GABAAR β3 subunit. In particular, α-Bungarotoxin blocks GABAARs that contain interfaces between adjacent β3 subunits5.
Labeled α-Bungarotoxin is ideally used to label nAChR α7 receptors, and in many cases to localize neuromuscular junctions6,7.
Alomone Labs is pleased to offer α-Bungarotoxin-ATTO Fluor-488 (#B-100-AG), labeled with a green fluorescent dye and α-Bungarotoxin-Biotin (#B-100-B), labeled with biotin.

Whole mount staining of mice Gastrocnemius muscle was stained with the neuromuscular junction marker α-Bungarotoxin-ATTO Fluor-488 (#B-100-AG), (green) at 1:50 (A) and 1:100 (B) ratios.
The images were taken using Nikon Epifluorescence microscopy at X100 magnification and are a kind gift from Dr. Eran Perlsson, Dept. of Physiology and Pharmacology, Tel-Aviv University.
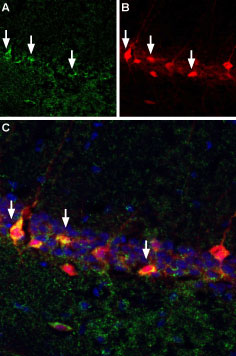
A. Free floating mouse brain sections were incubated with α-Bungarotoxin-Biotin (#B-100-B), (1:10,000) followed by streptavidin-Alexa 488 (green). B. Same sections were stained with antiparvalbumin, followed by goat anti-mouse labeled with Texas red isothiocyanate (TRITC). C. Merge of A and B demonstrates sites of colocalization (vertical arrows) confirming reports that a subset of hippocampal interneurons express nAChR α7. DAPI is used as the counterstain.
References
- Ohta, M. et al. (1987) FEBS Lett. 222, 79.
- Wilson, P.T. et al. (1988) Mol. Pharmacol. 34, 643.
- Wilson, S.P. and Kirshner, N. (1977) J. Neurochem. 28, 687.
- Garcia-Guzman, M. et al. (1995) Eur. J. Neurosci. 7, 647.
- McCann, C.M. et al. (2006) Proc. Natl. Acad. Sci. U.S.A. 103, 5149.
- Fertuck, H.C. and Salpeter, M.M. (1974) Proc. Natl. Acad. Sci. U.S.A. 71, 1376.
- Ravdin, P. and Axelrod, D. (1977) Anal. Biochem. 80, 585.