Overview
Cat #:
C-850
Alternative Name Actidione, Naramycin A
Lyophilized Powder yes
Origin Streptomyces griseus.
Source Natural
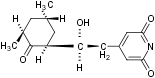
MW: 281.3
Purity: >98%
Effective concentration 10-100 µM.
Chemical name 4-[2-(3,5-Dimethyl-2-oxo-cyclohexyl)-2-hydroxyethyl]-2,6-piperidinedione.
Molecular formula C15H23NO4.
CAS No.: 66-81-9
Activity Cycloheximide is an antibiotic which inhibits protein synthesis in eukaryotes, but not in prokaryotes, by blocking translation elongation1,2.
References-Activity
- Bennett, L.L. Jr. et al. (1965) Biochim. Biophys. Acta 103, 478.
- Lin, W.W. and Hsu, Y.W. (2000) Cell Signal. 12, 457.
Shipping and storage Shipped at room temperature. Product as supplied can be stored intact at room temperature for several weeks. For longer periods, it should be stored at -20°C.
Solubility Water, DMSO, ethanol, methanol, acetone. Centrifuge all product preparations before use (10000 x g 5 min).
Storage of solutions Up to four weeks at 4°C or three months at -20°C.
Our bioassay
References - Scientific background
- Dewitte-Orr, S.J. et al. (2005) Fish Shellfish Immunol. 18, 279.
- Wool, I.G. and Cavicchi, P. (1966) Proc. Natl. Acad. U.S.A. 56, 991.
- Lin, W.W. and Hsu, Y.W. (2000) Cell Signal. 12, 457.
- Marcus, P.I. and Salb, J.M. (1966) Cold Spring Harb. Symp. Quant. Biol. 31, 335.
- Georg, L.K. (1953) AMA Arch. Derm. Syphilol. 67, 355.
- Kaufer N.F. et al. (1983) Nucleic Acids Res. 11, 3123.
Scientific background Cycloheximide is an antibiotic which is primarily used for the isolation of dermatophytes from clinical materials.1,2 It induces death of eukaryotic cells in an apoptosis-independent pathway. Cycloheximide acts as an elongation inhibitor during protein synthesis in eukaryotes, but not prokaryotes, by competing with the binding of ATP to the 60S ribosomal subunit. Mutations in the yeast gene CYH2 that encodes L29, a component of the 60S ribosomal subunit, can lead to cycloheximide resistance.3-6
Lyophilized Powder
Cycloheximide (#C-850) is a highly pure, natural, and biologically active compound.
For research purposes only, not for human use
Last Update: 02/01/2024
Applications
Citations
Citations