The brain is a web of complex neural circuits, made up of excitatory pyramidal neurons (PNs) and inhibitory interneurons (INs). While our understanding of how these circuits develop has advanced, excitatory neurons have typically been in the spotlight, while INs, despite their roles in neural functioning and their implication in diseases like epilepsy and schizophrenia, have been less explored in terms of development. However, recent paper published in Science Advances, has given them some much needed attention, and illuminates a particularly overlooked area: how neuromodulatory inputs like acetylcholine (ACh) influence the wiring of inhibitory neurons in the neocortex during early development.
A Focus on Chandelier Cells and the Cholinergic System
Researchers focussed on a specific type of inhibitory neuron called the chandelier cell (ChC). These neurons are known for their unique morphology and specialized role in controlling the firing of excitatory neurons. ChCs specifically regulate axon initial segments, which are vital for the generation of neural spikes, the brain’s communication signals. In this recent study, the authors elucidated how ACh, released from basal forebrain (BF) cholinergic neurons, impacts the development of these ChCs.
What the scientists found was fascinating: cholinergic inputs from the BF directly modulate ChC growth via nicotinic acetylcholine receptors (nAChRs) and T-type voltage-dependent calcium channels (VDCCs). These receptors and channels were found to regulate axonal filopodia, the precursors to axonal branches, independent of action potential generation. This indicates a direct, localized effect of ACh on the axonal arborization of ChCs.
Filopodia Initiation: A Key Event
A particularly compelling aspect of the research was the focus on filopodia initiation. Using in vivo imaging, researchers demonstrated that these slender structures indeed serve as the stepping stones for future branching. Through the activation of the nAChR-T-type VDCC pathway, ACh signaling influences the very initiation of these filopodia, thereby determining the eventual complexity of axonal branching.
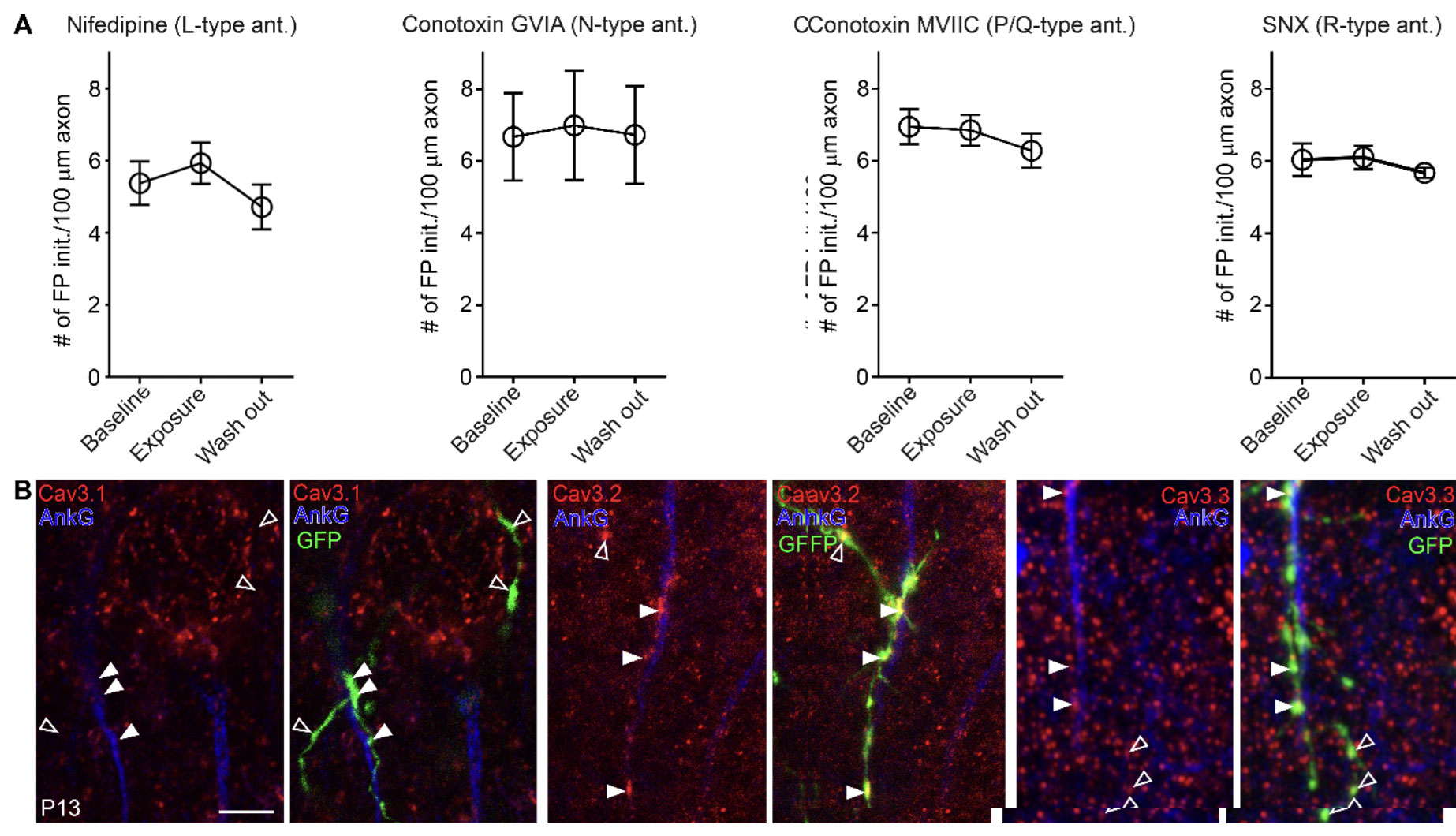
Among the intriguing findings is the identification of the specific VDCC isoforms involved in this process. Researchers initially posited that T-type VDCCs might be localized in axonal varicosities, the points where filopodia initiation preferentially occurs. To validate this, they examined the localization of T-type VDCC isoforms – CaV3.1, CaV3.2, and CaV3.3 – in endogenous ChCs using highly specific antibodies from Alomone Labs against these isoforms.
The results were illuminating: CaV3.2 and CaV3.3 were found to be localized in axonal varicosities, whereas CaV3.1 was barely present in ChC axons (Figure 1). This clearly demonstrates the specific isoforms acting at these critical points, possibly mediating the local calcium influx necessary for filopodia initiation.
Only some CaV isoforms are expressed in T-type VDCCs
Local vs. Systemic Impact and Fine Tuning
While it might be tempting to think that acetylcholine acts indirectly by modulating the activity of surrounding neurons, the study presents multiple lines of evidence to refute this. Inhibition of action potentials in the neurons under study didn’t alter filopodia initiation, suggesting that ACh’s effects are direct and local to the axons of ChCs. This rules out the possibility that changes in general synaptic transmission or spikes in surrounding neurons indirectly influence axonal arborization.
The research suggests that the degree of axonal branching in ChCs might be subject to fine-tuning based on the activity levels of basal forebrain cholinergic neurons. The implication here is fascinating: early-life experiences and environmental conditions could impact these activity levels, potentially leading to long-term consequences in how PNs generate spikes and process information.
Understanding the Etiology of Brain Disorders
While this research focused on the development of a specific inhibitory neuron, the chandelier cell, its findings could have broader implications. Disorders like schizophrenia, epilepsy, and ADHD have been linked to defects in both cholinergic signaling and inhibitory circuitry. This study provides a potential connection between these two lines of research, offering a fresh avenue for understanding the origins of such disorders.
A New Perspective on Cortical Circuit Assembly
The research illuminates a previously understudied aspect of brain development, showing how the cholinergic system intricately shapes inhibitory networks in the neocortex. It also opens up questions about how early-life experiences could influence this process, and how disruptions in it could be linked to various brain disorders. Clearly, understanding the role of the cholinergic system in the formation of inhibitory networks is more than an academic exercise – it’s a key piece in the puzzle of how the brain assembles itself.
Antibodies
- Anti-CACNA1G (CaV3.1) Antibody (#ACC-021)
- Anti-CaV3.2 (CACNA1H) Antibody (#ACC-025)
- Anti-CaV3.3 (CACNA1I) Antibody (#ACC-009)
Research Packs
The Research Packs contains all you need for T-Type CaV research: Antibodies for each T-type CaV channel and specific T-type CaV blockers
- T-Type CaV Channel Basic Research Pack (#ESB-101)
- T-Type CaV Channel Premium Research Pack (#ESP-101)
- T-Type CaV Channel Deluxe Research Pack (#ESD-101)

