Overview
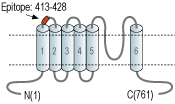
- Peptide (C)HQPSLDQPAIPSSKAT, corresponding to amino acid residues 413-428 of rat TRPV2 (Accession Q9WUD2). 1st extracellular loop.
Human - will not recognize the human epitope.
 Western blot analysis of rat brain (lanes 1 and 3) and rat RBL basophilic leukemia cell lysate (lanes 2 and 4):1,2. Guinea pig Anti-TRPV2 (VRL1) (extracellular) Antibody (#ACC-039-GP), (1:2000).
Western blot analysis of rat brain (lanes 1 and 3) and rat RBL basophilic leukemia cell lysate (lanes 2 and 4):1,2. Guinea pig Anti-TRPV2 (VRL1) (extracellular) Antibody (#ACC-039-GP), (1:2000).
3,4. Guinea pig Anti-TRPV2 (VRL1) (extracellular) Antibody, preincubated with TRPV2/VRL1 (extracellular) Blocking Peptide (#BLP-CC039).
 Multiplex staining of TRPV2 and mGluR5 in rat DRGImmunohistochemistry of rat dorsal root ganglion using Guinea pig Anti-TRPV2 (VRL1) (extracellular) Antibody (#ACC-039-GP) (1:60) and Anti-mGluR5 (extracellular)-ATTO Fluor-594 Antibody (#AGC-007-AR), (red), (1:60). A. TRPV2 staining (green). B. mGluR5 staining (red). C. Merge of A and B demonstrates co-localization of TRPV2 and mGluR5 in DRG cells. Nuclei are stained using DAPI as the counterstain (blue).
Multiplex staining of TRPV2 and mGluR5 in rat DRGImmunohistochemistry of rat dorsal root ganglion using Guinea pig Anti-TRPV2 (VRL1) (extracellular) Antibody (#ACC-039-GP) (1:60) and Anti-mGluR5 (extracellular)-ATTO Fluor-594 Antibody (#AGC-007-AR), (red), (1:60). A. TRPV2 staining (green). B. mGluR5 staining (red). C. Merge of A and B demonstrates co-localization of TRPV2 and mGluR5 in DRG cells. Nuclei are stained using DAPI as the counterstain (blue).
 Expression of TRPV2 in rat RBL cellsCell surface detection of TRPV2 in intact living rat basophilic leukemia (RBL) cells. A. Extracellular staining of cells using Guinea pig Anti-TRPV2 (VRL1) (extracellular) Antibody (#ACC-039-GP), (1:100) followed by goat anti-guinea pig-AlexaFluor-488 secondary antibody (green). B. Live view of the cells. C. Merge image of A and B.
Expression of TRPV2 in rat RBL cellsCell surface detection of TRPV2 in intact living rat basophilic leukemia (RBL) cells. A. Extracellular staining of cells using Guinea pig Anti-TRPV2 (VRL1) (extracellular) Antibody (#ACC-039-GP), (1:100) followed by goat anti-guinea pig-AlexaFluor-488 secondary antibody (green). B. Live view of the cells. C. Merge image of A and B.
- Montell, C. et al. (2002) Mol. Cell 9, 229.
- Clapham, D.E. (2003) Nature 426, 517.
- Moran, M.M. et al. (2004) Curr. Opin. Neurobiol. 14, 362.
- Clapham, D.E. et al. (2003) Pharmacol. Rev. 55, 591.
- Gunthorpe, M.J. et al. (2002) Trends Pharmacol. Sci. 23, 183.
- Tominaga, M. and Caterina, M.J. et al. (2004) J. Neurobiol. 61, 3.
- Voets, T. et al. (2004) Nature 430, 748.
- Pedersen, S.F. et al. (2005) Cell Calcium 38, 233.
- Muraki, K. et al. (2003) Circ. Res. 93, 829.
- Shimosato, G. et al. (2005) Pain 119, 225.
- Saunders, C.I. et al. (2007) Mol. Immunol. 44, 1429.
- Caterina, M.J. et al. (1999) Nature 398, 436.
TRP channels are a large family (about 28 genes) of plasma membrane, non-selective cationic channels that are either specifically or ubiquitously expressed in excitable and non-excitable cells.1 The TRP channels have six putative transmembrane domains (TM) with a pore domain between the fifth and the sixth TM, and all assemble as tetramers. Both the N- and the C-termini of all TRPs are intracellular3.
According to IUPHAR, the TRP family is comprised of numerous subfamilies on the basis of sequence homology; TRPC, TRPM, TRPV, TRPA, TRPML, and TRPP1-4. The TRPV subfamily consists of six members, TRPV1-65.
Four members of the TRPV family have been described as thermosensitive ion channels (TRPV1 to TRPV4). Each channel exhibits distinct thermal activation thresholds ranging from noxious cold (<17°C) to noxious heat (>52°C) 6,7. Although it shares around 50% homology with TRPV1, TRPV2 is not activated by capsaicin nor by protons. It has a high temperature threshold of ~52°C and is considered to play an essential role in the perception of high-intensity noxious heat stimulation8-10. TRPV2 is also considered to be a stretch-activated channel and to play a role in skeletal and cardiac muscle degeneration and pain pathway8. The TRPV2 channel is expressed in DRG neurons, different brain regions and non-neuronal tissues such as the spleen, lung and intestine and in components of the immune system5,11,12.
Application key:
Species reactivity key:
Alomone Labs is pleased to offer a highly specific antibody directed against an epitope of rat TRPV2. Guinea pig Anti-TRPV2 (VRL1) (extracellular) Antibody (#ACC-039-GP) raised in guinea pigs, can be used in western blot and immunocytochemistry. It has been designed to recognize TRPV2 from mouse and rat samples. The antigen used to immunize guinea pigs is the same as Anti-TRPV2 (VRL1) (extracellular) Antibody (#ACC-039) raised in rabbit. Our line of guinea pig antibodies enables more flexibility with our products such as multiplex staining studies, immunoprecipitation, etc.

Multiplex staining of SERCA1 and TRPV2 in mouse heart.Immunohistochemical staining of mouse heart immersion-fixed, free floating frozen sections, using rabbit Anti-SERCA1 Antibody (#ACP-011) (1:200) and Guinea pig Anti-TRPV2 (VRL1) (extracellular) Antibody (#ACC-039-GP), (1:200). A. SERCA1 staining (green) appears in T tubules (arrows). B. TRPV2 staining (red) in same section is detected in intercalated discs (diagonal arrows) and T tubules (horizontal arrow). C. Merge of panels A and B demonstrates co-localization of SERCA1 and TRPV2. Nuclei are stained using DAPI (blue).
Applications
Citations
- Mouse BV-2 microglia lysate.
Maksoud, M.J.E. et al. (2019) Glia 67, 2294.
- Mouse BV-2 microglia.
Maksoud, M.J.E. et al. (2019) Glia 67, 2294.
- Mouse BV-2 microglia.
Maksoud, M.J.E. et al. (2019) Glia 67, 2294.
