Overview
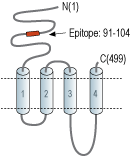
- Peptide (C)KWKPSDYQGVEFMR, corresponding to amino acid residues 91-104 of rat nAChRα3 (Accession P04757). Extracellular, N-terminus.
 Western blot analysis of rat brain membranes:1. Anti-Nicotinic Acetylcholine Receptor α3 (CHRNA3) (extracellular) Antibody (#ANC-003), (1:200).
Western blot analysis of rat brain membranes:1. Anti-Nicotinic Acetylcholine Receptor α3 (CHRNA3) (extracellular) Antibody (#ANC-003), (1:200).
2. Anti-Nicotinic Acetylcholine Receptor α3 (CHRNA3) (extracellular) Antibody, preincubated with Nicotinic Acetylcholine Receptor α3/CHRNA3 (extracellular) Blocking Peptide (#BLP-NC003).
 Expression of Nicotinic acetylcholine receptor α3 in rat neocortexImmunohistochemical staining of immersion-fixed, free floating rat brain frozen sections were stained using Anti-Nicotinic Acetylcholine Receptor α3 (CHRNA3) (extracellular) Antibody (#ANC-003), (1:200). A. Intense staining (green) of nAChR α3 appears in apical dendrites (thin arrows). A few cortical interneurons (thick arrow) are also stained with the antibody. B. The same section was stained also for parvalbumin (red staining). An arrow depicts a neuron expressing both nAChR α3 and parvalbumin.
Expression of Nicotinic acetylcholine receptor α3 in rat neocortexImmunohistochemical staining of immersion-fixed, free floating rat brain frozen sections were stained using Anti-Nicotinic Acetylcholine Receptor α3 (CHRNA3) (extracellular) Antibody (#ANC-003), (1:200). A. Intense staining (green) of nAChR α3 appears in apical dendrites (thin arrows). A few cortical interneurons (thick arrow) are also stained with the antibody. B. The same section was stained also for parvalbumin (red staining). An arrow depicts a neuron expressing both nAChR α3 and parvalbumin.
- Albuquerque, E.X. et al. (2009) Physiol. Rev. 89, 73.
- Karlin, A. et al. (1986) Ann. NY. Acad. Sci. 463, 53.
- Kalamida, D. et al. (2007) FEBS J. 274, 3799.
- Vernino, S. et al. (2009) Auton. Neurosci. 146, 3.
- Xu, W. et al. (1999) Proc. Natl. Acad. Sci. U.S.A. 96, 5746.
- Young, T. et al. (2008) Brain Res. 1229, 118.
Acetylcholine, released by cholinergic neurons, activates two groups of acetylcholine receptors (AChRs); muscarinic AChRs (mAChRs) which belong to the superfamily of G-protein coupled receptors (GPCRs) and nicotinic AChRs (nAChRs) which belong to the ligand-gated ion channel superfamily. nAChRs also respond to nicotine, hence their name1.
To date, 17 different but related subunits of nAChRs have been identified and cloned. They consist of a subunits (α1-10), which is responsible for the binding of ligands. In fact, this subunit includes a Cys-loop in the first extracellular domain that is required for agonist binding2. The other subunits responsible for making up the active receptor are the β (β1-4), γ, δ and ε subunits3. Structurally, all subunits have the following: a conserved large extracellular N-terminal domain, 3 conserved transmembrane domains, a variable cytoplasmic loop and a fourth transmembrane domain with a short extracellular C-terminal domain. An active nAChR is generally a heteropentamer of these various subunits organized around a central pore1.
All α subunits are expressed in neuronal cells except for the α1 subunit which is specifically expressed in the muscle3. In the peripheral autonomic nervous system, nicotinic receptors are prominent and are responsible mediating fast synaptic transmission in all peripheral autonomic ganglia4. In this system, these channel receptors are mostly made up of two α3 subunits heteromerized with three other subunits. The α3 subunit plays a most important role in this system as knockout mice suffer from severe autonomic failure accompanied by gastrointestinal and bladder malfunctions5.
Autoimmune autonomic ganglionopathy (AAG) is a neurological disorder characterized by dysfunctions in sympathetic, parasympathetic and enteric functions. Patients suffering from the disease express high levels of self antibodies against ionotropic nicotinic receptors, namely α3 subunits4. The α3 subunit is also found in dorsal root ganglion where it is most probably involved in nociception6.
Application key:
Species reactivity key:
Alomone Labs is pleased to offer a highly specific antibody directed against an epitope located at the extracellular N-terminal domain of the rat nAChRα3. Anti-Nicotinic Acetylcholine Receptor α3 (CHRNA3) (extracellular) Antibody (#ANC-003) can be used in western blot and immunohistochemistry applications. It has been designed to recognize nAChRα3 from mouse, rat and human samples.
