Overview
- Osteen, J.D. et al. (2016) Nature 534, 494.
 Alomone Labs Hm1a Toxin enhances the current of NaV1.1 channels expressed in Xenopus oocytes.A. Representative time course of Hm1a Toxin (#STH-601) effect on the normalized area of NaV1.1 channel current. Membrane potential was held at -90 mV, current was elicited by a 100 ms voltage step to -10 mV every 10 sec and was significantly enhanced by the application of 250 nM Hm1a Toxin (green). B. Superimposed traces of NaV1.1 current after application of control (black) and of 250 nM Hm1a Toxin (green), taken from the recording in A.
Alomone Labs Hm1a Toxin enhances the current of NaV1.1 channels expressed in Xenopus oocytes.A. Representative time course of Hm1a Toxin (#STH-601) effect on the normalized area of NaV1.1 channel current. Membrane potential was held at -90 mV, current was elicited by a 100 ms voltage step to -10 mV every 10 sec and was significantly enhanced by the application of 250 nM Hm1a Toxin (green). B. Superimposed traces of NaV1.1 current after application of control (black) and of 250 nM Hm1a Toxin (green), taken from the recording in A.
- Escoubas, P. et al. (2002) Mol. Pharmacol. 62, 48.
- Osteen, J.D. et al. (2016) Nature 534, 494.
- Osteen, J.D. et al. (2017) Proc. Natl. Acad. Sci. U.S.A. 114, 6836.
Hm1a Toxin (δ-TRTX-Hm1a) is a peptide toxin originally isolated from Heteroscodra maculate tarantula venom. The toxin was originally described as a KV2 and KV4 channel blocker1 but has since been found to be a selective and specific activator of NaV1.1 voltage-gated sodium channels2,3. It interacts with extracellular loops connecting transmembrane segments 1-2 and 3-4 in domain IV voltage sensor of the channel to inhibit NaV1.1 fast inactivation2,3. Hm1a inhibits human NaV1.1 channel inactivation expressed in Xenopus oocytes with EC50 value of 38 ± 6 nM.
NaV1.1 channel is a therapeutic target for brain disorders, such as epilepsy, Alzheimer's disease, and autism. It also contributes to mechanical pain by regulating excitability in a specific subset of sensory neurons within the peripheral nervous system.
Hm1a Toxin (#STH-601) is a highly pure, synthetic, and biologically active peptide toxin.
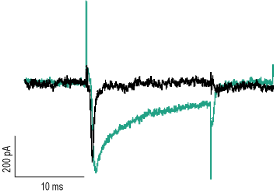
Alomone Labs Hm1a Toxin enhances the current of NaV1.1 channels transiently expressed in HEK293 cells.Representative superimposed traces of NaV1.1 currents in the absence (black) or presence of 250 nM Hm1a Toxin (#STH-601), (green). Currents were elicited by a 20 ms voltage step to 0 mV every 10 sec from a holding potential of -120 mV.This figure was kindly provided by the lab of Dr. Moran Rubinstein, Goldschleger Eye Research Institute, The Department of Human Molecular Genetics and Biochemistry, Sackler School of Medicine, Tel Aviv University.
Applications
Citations
- HEK-293 cells expressing NaV1.1 channel.
Nissenkorn, A. et al. (2019) PLoS ONE 14, e0211901.

