Overview
- Peptide CKALHVLFERVSEE, corresponding to amino acid residues 183-196 of mouse LRRK2 (Accession Q5S006). N-terminus.

 Western blot analysis of rat (lanes 1 and 3) and mouse (lanes 2 and 4) brain membranes:1,2. Anti-LRRK2 Antibody (#ANR-102), (1:200).
Western blot analysis of rat (lanes 1 and 3) and mouse (lanes 2 and 4) brain membranes:1,2. Anti-LRRK2 Antibody (#ANR-102), (1:200).
3,4. Anti-LRRK2 Antibody, preincubated with LRRK2 Blocking Peptide (#BLP-NR102).
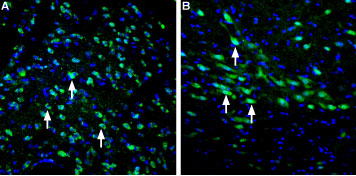 Expression of LRRK2 in rat striatum and substantia nigraImmunohistochemical staining of perfusion-fixed frozen rat brain sections using Anti-LRRK2 Antibody (#ANR-102), (1:400), followed by goat-anti-rabbit-AlexaFluor-488 secondary antibody. A. LRKK2 staining (green) of striatum sections is detected in the cytoplasm of several striatal cells (arrows). B. Staining in the substantia nigra pars compacta shows LRKK2 staining (green) in nuclei and cytoplasm of several cells (arrows). Nuclei were stained with DAPI (blue).
Expression of LRRK2 in rat striatum and substantia nigraImmunohistochemical staining of perfusion-fixed frozen rat brain sections using Anti-LRRK2 Antibody (#ANR-102), (1:400), followed by goat-anti-rabbit-AlexaFluor-488 secondary antibody. A. LRKK2 staining (green) of striatum sections is detected in the cytoplasm of several striatal cells (arrows). B. Staining in the substantia nigra pars compacta shows LRKK2 staining (green) in nuclei and cytoplasm of several cells (arrows). Nuclei were stained with DAPI (blue).
- Seol, W. (2010) BMB Reports 43, 233.
Parkinson’s disease (PD) is the second most common neurodegenerative disease. The condition is inherited in 5-10% of all PD patients. Mutations in the PARK8 gene have been implicated in the development of PD.
PARK8 gene encodes the protein LRRK2 (Leucine-rich repeat kinase 2). It is a multi-domain protein containing distinctive enzymatic domains of GTPase and a kinase, the protein interaction motifs LRR (Leucine-rich repeat) and WD40. LRRK2’s domain structure indicates that it belongs to the Roco protein family. LRRK2 is expressed in the kidney, lung and dopaminergic neurons in the substantia nigra. It is mostly located in the cytosol, however, it is also expressed on membranes of organelles such as the mitochondria, golgi apparatus and synaptic vesicles.
There are several PARK8 mutations that have been associated with both familial and sporadic PD. The most common mutation, G2019S, is present in 85% of PD patients carrying PARK8 mutations and in 4% of all familial PD patients. In addition, this mutation has been identified in 1% of sporadic PD patients. The prevalence of this mutation is especially high in Ashkenazi Jewish and North-African Arab populations. This mutation causes increased kinase activity while other mutations, such as R1441C, decrease GTPase activity. Overexpression of the LRRK2 protein causes neuronal cell death and other pathological conditions which are aggravated by the overexpression of mutant proteins.
The exact role of LRRK2 in PD pathophysiology still remains unclear1.
Application key:
Species reactivity key:
Anti-LRRK2 Antibody (#ANR-102) is a highly specific antibody directed against an epitope of the mouse protein. The antibody can be used in western blot and immunohistochemistry applications. It has been designed to recognize LRKK2 from human, mouse, and rat samples.
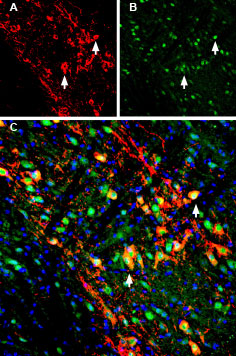 Multiplex staining of GIRK2 (Kir3.2) and LRRK2 in mouse substantia nigra pars compacta (SNC).Immunohistochemical staining of immersion-fixed, free floating mouse brain frozen sections using Guinea pig Anti-GIRK2 (Kir3.2) Antibody (#APC-006-GP), (1:200) and rabbit Anti-LRRK2 Antibody (#ANR-102), (1:200). A. Kir3.2 staining (red) appears in profiles of dopaminergic neurons (arrows). B. LRRK2 staining (green) is detected in several cell bodies in the SNC. C. Merge of the two images demonstrates colocalization in several neurons (arrows). Cell nuclei are stained with DAPI (blue).
Multiplex staining of GIRK2 (Kir3.2) and LRRK2 in mouse substantia nigra pars compacta (SNC).Immunohistochemical staining of immersion-fixed, free floating mouse brain frozen sections using Guinea pig Anti-GIRK2 (Kir3.2) Antibody (#APC-006-GP), (1:200) and rabbit Anti-LRRK2 Antibody (#ANR-102), (1:200). A. Kir3.2 staining (red) appears in profiles of dopaminergic neurons (arrows). B. LRRK2 staining (green) is detected in several cell bodies in the SNC. C. Merge of the two images demonstrates colocalization in several neurons (arrows). Cell nuclei are stained with DAPI (blue).
