Overview
- Jin, W. and Lu, Z. (1998) Biochemistry 37, 13291.
- Jin, W. et al. (1999) Biochemistry 38, 14294.
- Drici, M.D. et al. (2000) Br. J. Pharmacol. 131, 569.
- Kitamura, H. et al. (2000) Pharmacol. Exp. Ther. 293, 196.
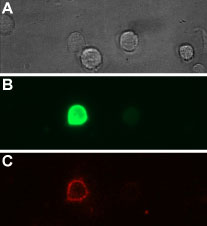 Alomone Labs Tertiapin-Q-ATTO Fluor-633 binds GIRK1/GIRK4-GFP transfected HEK293T cells.Living cells were incubated in the presence of 100 nM Tertiapin-Q-ATTO Fluor-633 (#STT-170-FR). A. Visible light of cells. B. GIRK1/GIRK4-GFP transfected cells (green). C. Tertiapin-Q-ATTO Fluor-633 (red).
Alomone Labs Tertiapin-Q-ATTO Fluor-633 binds GIRK1/GIRK4-GFP transfected HEK293T cells.Living cells were incubated in the presence of 100 nM Tertiapin-Q-ATTO Fluor-633 (#STT-170-FR). A. Visible light of cells. B. GIRK1/GIRK4-GFP transfected cells (green). C. Tertiapin-Q-ATTO Fluor-633 (red).
The pictures are a kind gift from the lab of Prof. Eithan Reuveny, Weizmann Institute, Israel. Alomone Labs Tertiapin-Q-ATTO Fluor-633 inhibits Kir3.2 channel heterologously expressed in Xenopus oocytes.A continuous current trace recorded at a holding potential of -80 mV. Kir3.2 currents are downward reflections activated by high K+ containing solution. While activated, 50 nM and 100 nM of Tertiapin-Q-ATTO Fluor-633 (#STT-170-FR) were applied for 2 min (indicated as bars).
Alomone Labs Tertiapin-Q-ATTO Fluor-633 inhibits Kir3.2 channel heterologously expressed in Xenopus oocytes.A continuous current trace recorded at a holding potential of -80 mV. Kir3.2 currents are downward reflections activated by high K+ containing solution. While activated, 50 nM and 100 nM of Tertiapin-Q-ATTO Fluor-633 (#STT-170-FR) were applied for 2 min (indicated as bars).
Tertiapin, the native toxin, was originally isolated from European honey bee Apis mellifera venom. Native and synthetic Tertiapin blocks a range of inward rectifier K+ channels (Kir), in particular ROMK1 (Kir1.1, IC50 = 2 nM) and GIRK (Kir3 family, IC50 for the Kir3.1/3.4 heteromer was 8.6 nM) but with no effect on the Kir2 family member1. In accordance, it was shown to inhibit acetylcholine induced K+ currents in mammalian cardiomyocytes2,3.
Tertiapin-Q is a derivative of Tertiapin in which Met13 is substituted by a Gln residue. However, unlike native Tertiapin, Tertiapin-Q is non-oxidizable and therefore is more stable4.
Tertiapin-Q inhibits the above-mentioned channels with similar affinities and also inhibits Ca2-activated large conductance BK-type K+ channels in a concentration and voltage-dependent manner5.

