Overview
- Carter, R.B. et. al. (1997) J. Pharmacol. Exp. Ther. 280, 1284.
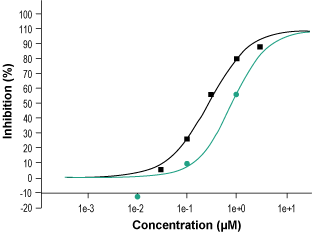 Alomone Labs Ganaxolone inhibits specific binding of TBPS to rat brain cortex GABA(A) receptors.Percent inhibition of specific binding of 2 nM [35S] TBPS to membranes from rat brain cerebral cortex was plotted against concentrations of Ganaxolone (#G-120), (green circles) and of Picrotoxin (assay standard, black squares) and used to calculate IC50 and Ki values for Ganaxolone (IC50 ≈ 0.81 µM, and Ki ≈ 0.79 µM).
Alomone Labs Ganaxolone inhibits specific binding of TBPS to rat brain cortex GABA(A) receptors.Percent inhibition of specific binding of 2 nM [35S] TBPS to membranes from rat brain cerebral cortex was plotted against concentrations of Ganaxolone (#G-120), (green circles) and of Picrotoxin (assay standard, black squares) and used to calculate IC50 and Ki values for Ganaxolone (IC50 ≈ 0.81 µM, and Ki ≈ 0.79 µM).
- Carter, R.B. et. al. (1997) J. Pharmacol. Exp. Ther. 280, 1284.
- Nohria, V. and Giller, E. (2007) Neurotherapeutics 4, 102.
- Reddy, D.S. and Rogawski, M.A. (2010) Epilepsy Res. 89, 254.
Ganaxolone (CCD 1042) is a 3β-methyl-substituted synthetic analogue of the endogenous neuroactive steroid allopregnanolone with anticonvulsant, anxiolytic, and sedative effects. The methyl group on the compound makes it more resistant to metabolism and increases the compound’s half life by hours.
Ganaxolone is a selective, potent and positive allosteric modulator of the GABA(A) receptors. It enhances the binding of the benzodiazepine and the GABA sites. In several clinical studies, ganaxolone treatment showed to have protective activity in several seizures models in adults and in children with history of infantile spasms. In adults the compound successfully reduced the frequency of partial seizures2,3.
Ionotropic GABA(A) receptors mediate GABA action, a major inhibitory neurotransmitter, through the central nervous system. The receptors are considered to be a major target of antiseizure drugs1,2.
Ganaxolone (#G-120) is a highly pure, synthetic, and biologically active compound.