Overview
Cat #:
STG-250
Alternative Name β-theraphotoxin-Gr1a, β-TRTX-Gr1a
Lyophilized Powder yes
Origin Synthetic peptide
MW: 3697 Da
Purity: >98% (HPLC)
Effective concentration 20-500 nM.
Sequence YCQKWMWTCDSKRKCCEDMVCQLWCKKRL.
Modifications Disulfide bonds between Cys2-Cys16, Cys9-Cys21 and Cys15-Cys25.
Molecular formula C159H243N45O41S8.
Activity GrTx1 is a blocker of voltage-gated Na+ channels and herg1 channels1.
References-Activity
- Redaelli, E. et al. (2010) J. Biol. Chem. 285, 4130.
Accession number P85117.
Shipping and storage Shipped at room temperature. Product as supplied can be stored intact at room temperature for several weeks. For longer periods, it should be stored at -20°C.
Solubility Any other aqueous buffer. Centrifuge all product preparations before use (10000 x g 5 min).
Storage of solutions Up to two weeks at 4°C or three months at -20°C.
Our bioassay
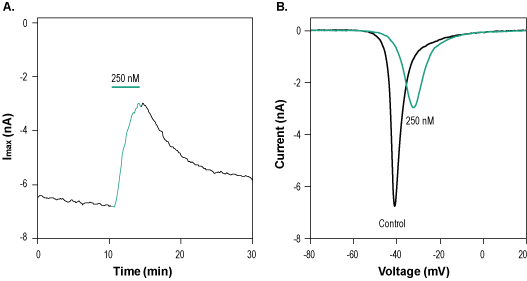 Alomone Labs GrTx1 inhibits NaV1.7 currents in stably transfected HEK293T cells.A. Time course of GrTx1 (#STG-250) action on stably transfected NaV1.7 currents. Current amplitudes were plotted as a function of time. Membrane potential was held at -100 mV and currents were elicited by a 100 ms voltage ramp from the holding potential of -100 mV to +60 mV, applied every 10 sec using whole-cell voltage clamp. 250 nM GrTx1 were perfused as indicated by the green horizontal bar. B. Superimposed traces of Nav1.7 channel currents in the absence (control) and presence (green) of 250 nM GrTx1 applied for 2.5 min. (taken from the experiment in A).
Alomone Labs GrTx1 inhibits NaV1.7 currents in stably transfected HEK293T cells.A. Time course of GrTx1 (#STG-250) action on stably transfected NaV1.7 currents. Current amplitudes were plotted as a function of time. Membrane potential was held at -100 mV and currents were elicited by a 100 ms voltage ramp from the holding potential of -100 mV to +60 mV, applied every 10 sec using whole-cell voltage clamp. 250 nM GrTx1 were perfused as indicated by the green horizontal bar. B. Superimposed traces of Nav1.7 channel currents in the absence (control) and presence (green) of 250 nM GrTx1 applied for 2.5 min. (taken from the experiment in A).
References - Scientific background
- Clement, H. et al. (2007) Toxicon 50, 65.
- Redaelli, E. et al. (2010) J. Biol. Chem. 285, 4130.
Scientific background β-theraphotoxin-Gr1a (GrTx1) is a peptidyl toxin originally isolated from the venom of the ‘‘rosean-tarantula’’ (Grammostola rosea). GrTx1 has 29 amino-acid residues, compactly folded by three disulfide bridges with a molecular weight of 3697 Da. Sequence analysis reveals that GrTx1 is closely related to other spider toxins reported to affect various distinct ion channel functions1. The toxin inhibits NaV1.1 (IC50 = 0.63 μM), NaV1.2 (IC50= 0.23 μM), NaV1.3 (IC50 = 0.77 μM), NaV1.4 (IC50= 1.29 μM), NaV1.6 (IC50= 0.63 μM), NaV1.7 (IC50= 0.37 μM) and potassium channels KV11.1 (IC50 = 1.2 μM)2.
Target NaV, KV11.1 channels
Peptide Content: 100%
Lyophilized Powder
GrTx1 (#STG-250) is a highly pure, synthetic, and biologically active peptide toxin.
For research purposes only, not for human use
Last Update: 07/05/2024
Applications
Citations
Citations

