Overview
- Ostrow, K.L. et al. (2003) Toxicon 42, 263.
- Suchyna, T.M. et al. (2000) J. Gen. Physiol. 115, 583.
- Redaelli, E. et al. (2010) J. Biol. Chem. 285, 4130.
 Alomone Labs GsMTx-4 inhibits NaV1.7 channel currents expressed in Xenopus oocytes.NaV1.7 currents were elicited by 100 ms voltage ramp from a holding potential of -100 mV to +30 mV, applied every 10 sec using whole-cell voltage clamp technique. Left: Superimposed traces of NaV1.7 currents before (black) and during (green) application of 500 nM GsMTx-4 (#STG-100). Right: GsMTx-4 dose response inhibition of NaV1.7 currents.
Alomone Labs GsMTx-4 inhibits NaV1.7 channel currents expressed in Xenopus oocytes.NaV1.7 currents were elicited by 100 ms voltage ramp from a holding potential of -100 mV to +30 mV, applied every 10 sec using whole-cell voltage clamp technique. Left: Superimposed traces of NaV1.7 currents before (black) and during (green) application of 500 nM GsMTx-4 (#STG-100). Right: GsMTx-4 dose response inhibition of NaV1.7 currents.
- Suchyna, T.M. et al. (2000) J. Gen. Physiol. 115, 583.
- Bode, F. et al. (2001) Nature 409, 35.
- Fang, J. and Iwasa, K.H. (2006) Neurosci. Lett. 404, 213.
- Redaelli, E. et al. (2010) J. Biol. Chem. 285, 4130.
- Spassova, M.A. et al. (2006) Proc. Natl. Acad. Sci. U.S.A. 103, 16586.
- Alessandri-Haber, N. et al. (2009) J. Neurosci. 29, 6217.
- Ostrow, K.L. et al. (2003) Toxicon 42, 263.
- Bae, C. et al. (2011) Biochemistry 50, 6295.
GsMTx-4 is a 34 amino acid peptidyl toxin originally isolated from the Grammostola rosea (Chilean rose) tarantula venom and belongs to the huwentoxin-1 family1.
This toxin inhibits different channels and in addition has antimicrobial activity. It blocks cation-selective mechanosensitive ion channels (strech-activated channels, SACs), without having an effect on whole-cell voltage-sensitive currents1. In addition, it inhibits atrial fibrillation2 as well as the membrane motor of outer hair cells3 at low doses. A medium toxicity on a large spectra of voltage-gated Na+ channels, namely NaV1.1, NaV1.2, NaV1.3, NaV1.4, NaV1.5, NaV1.6 and NaV1.7 was reported. GsMTx-4 also inhibits K+ channels KV11.1 and KV11.2, whereas it does not inhibit K+ channels KV1.1 (IC50 > 85 µM), KV1.4 (IC50 > 85 µM) and KV11.3 (IC50 = 53 µM)4. GsMTx-4 was also found to inhibit both TRPC1 and TRPC6 channels5,6, as well as Piezo1, the mechanosensitive channel7.
Antimicrobial activity is shown against several Gram-positive and Gram-negative bacteria as well8.
GsMTx-4 (#STG-100) is a highly pure, synthetic, and biologically active peptide toxin.
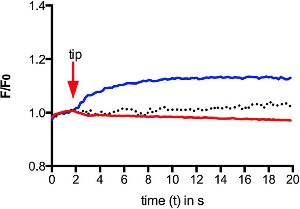
Alomone Labs GsMTx-4 inhibits mechanical-induced Ca2+ increase in human RBCs.Intracellular Ca2+ levels from Fluo-4 AM loaded human red blood cells (RBCs) were measured following mechanical stimulation touching a cell with a micropipette (blue). Application of 2.5 µM GsMTx-4 (#STG-100) prior to the stimulation blocked the intracellular Ca2+ increase (red). Control, without stimulation is depicted by the dotted graph.Adapted from Danielczok, J.G. et al. (2017) Front. Physiol. 8, 979. with permission of Frontiers.
Applications
Citations
 Inhibiting mechano-sensitive ion channels reduces valvular myofibroblast differentiation.Immunocytochemical staining of VICs treated with the mechano-sensitve ion channel inhibitor GsMTX (#STG-100) at 5 μM. αSMA protein expression was reduced relative to the internal control, GAPDH (not shown). Similarly, αSMA stress fibers (green) were inhibited by the inhibitor. Nuclei, blue. (Scale bars, 100 μm.).
Inhibiting mechano-sensitive ion channels reduces valvular myofibroblast differentiation.Immunocytochemical staining of VICs treated with the mechano-sensitve ion channel inhibitor GsMTX (#STG-100) at 5 μM. αSMA protein expression was reduced relative to the internal control, GAPDH (not shown). Similarly, αSMA stress fibers (green) were inhibited by the inhibitor. Nuclei, blue. (Scale bars, 100 μm.).
Adapted from Wang, H. et al. (2013) with permission of the National Academy of Sciences, USA.
- Mouse intraperitoneal injection.
Velasco-Estevez, M. et al. (2020) Glia 68, 356.
- Mouse stromal vascular cell fraction and adipocytes.
Zhao, C. et al. (2019) Front. Endocrinol. 10, 373.
- Danielczok, J.G. et al. (2017) Front. Physiol. 8, 979.
- Johansson, S.E. et al. (2015) Acta Physiol. 214, 376.
- Kim, J.C. and Woo, S.H. (2015) J. Physiol. 593, 5091.
- Lic, C. et al. (2015) Sci. Rep. 5, 8364.
- Vemana, H.P. et al. (2015) PLoS ONE 10, e0125764.
- Arora, P.D. et al. (2013) Mol. Biol. Cell. 24, 734.
- Wang, H. et al. (2013) Proc. Natl. Acad. Sci. U.S.A. 110, 19336.

