Overview
- Highly purified 2.5S mouse NGF (Accession P01139).
 NGF-induced neurite outgrowth neutralizing assayPC12 cells were grown on collagen-coated plates. A. In the absence of NGF. B. In the presence of Native mouse NGF 2.5S protein (99%) (#N-240) (50 ng/ml). C. In the presence of Native mouse NGF 2.5S protein (99%) (50 ng/ml) with Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) (1000 ng/ml). The development of neurites over a period of 6 days was visualized by Methylene Blue staining.
NGF-induced neurite outgrowth neutralizing assayPC12 cells were grown on collagen-coated plates. A. In the absence of NGF. B. In the presence of Native mouse NGF 2.5S protein (99%) (#N-240) (50 ng/ml). C. In the presence of Native mouse NGF 2.5S protein (99%) (50 ng/ml) with Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) (1000 ng/ml). The development of neurites over a period of 6 days was visualized by Methylene Blue staining. proNGF-induced neurite outgrowth neutralizing assayPC12 cells were grown on collagen-coated plates. A. In the absence of proNGF. B. In the presence of Recombinant mouse proNGF protein (#N-250) (100 ng/ml). C. In the presence of Recombinant mouse proNGF protein (100 ng/ml) with Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) (500 ng/ml). The development of neurites over a period of 6 days was visualized by Methylene Blue staining.
proNGF-induced neurite outgrowth neutralizing assayPC12 cells were grown on collagen-coated plates. A. In the absence of proNGF. B. In the presence of Recombinant mouse proNGF protein (#N-250) (100 ng/ml). C. In the presence of Recombinant mouse proNGF protein (100 ng/ml) with Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) (500 ng/ml). The development of neurites over a period of 6 days was visualized by Methylene Blue staining. Neutralization of NGF-mediated PC12 proliferationPC12 cells were grown in the absence of serum and in the presence of Native mouse NGF 2.5S protein (99%) (#N-240), (50 ng/ml) as indicated. Varying concentrations of Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) were added to the culture and cell viability was established after 24 h using the XTT method. Note that 600 ng/ml Mouse NGF/proNGF Neutralizing Antibody was able to completely block the activity of 50 ng/ml mouse NGF.
Neutralization of NGF-mediated PC12 proliferationPC12 cells were grown in the absence of serum and in the presence of Native mouse NGF 2.5S protein (99%) (#N-240), (50 ng/ml) as indicated. Varying concentrations of Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) were added to the culture and cell viability was established after 24 h using the XTT method. Note that 600 ng/ml Mouse NGF/proNGF Neutralizing Antibody was able to completely block the activity of 50 ng/ml mouse NGF.
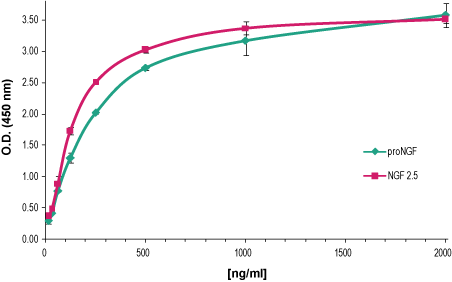 Indirect ELISA of adsorbed proNGF and NGFIncreasing amounts of Recombinant mouse proNGF protein (#N-250), (blue) and Native mouse NGF 2.5S protein (99%) (#N-240), (red) were adsorbed to a microplate and detected with Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) (2 μg/ml), followed by goat-anti-mouse-HRP and chromogenic substrate.
Indirect ELISA of adsorbed proNGF and NGFIncreasing amounts of Recombinant mouse proNGF protein (#N-250), (blue) and Native mouse NGF 2.5S protein (99%) (#N-240), (red) were adsorbed to a microplate and detected with Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) (2 μg/ml), followed by goat-anti-mouse-HRP and chromogenic substrate.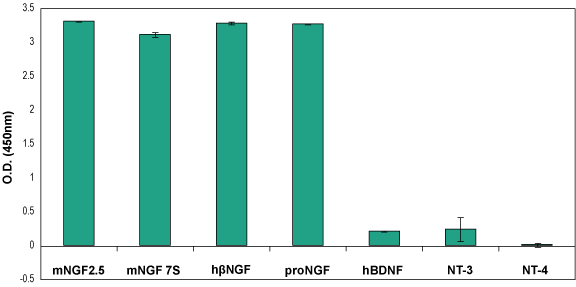 Indirect ELISA of adsorbed neurotrophic factorsNeurotrophic factors Native mouse NGF 2.5S protein (99%) (#N-240), Native mouse NGF 7S protein (#N-130), Recombinant human beta-NGF protein (#N-245), Recombinant human proNGF protein (#N-280), Recombinant human BDNF protein (#B-250), Recombinant human Neurotrophin-3 (NT-3) protein (#N-260) and Recombinant human Neurotrophin-4 (NT-4) protein (#N-270) all at 2 μg/ml were adsorbed to a microplate and analyzed using Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) (2 μg/ml), followed by goat-anti-mouse-HRP and chromogenic substrate.
Indirect ELISA of adsorbed neurotrophic factorsNeurotrophic factors Native mouse NGF 2.5S protein (99%) (#N-240), Native mouse NGF 7S protein (#N-130), Recombinant human beta-NGF protein (#N-245), Recombinant human proNGF protein (#N-280), Recombinant human BDNF protein (#B-250), Recombinant human Neurotrophin-3 (NT-3) protein (#N-260) and Recombinant human Neurotrophin-4 (NT-4) protein (#N-270) all at 2 μg/ml were adsorbed to a microplate and analyzed using Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) (2 μg/ml), followed by goat-anti-mouse-HRP and chromogenic substrate.
- Roux, P.P. et al. (2002) Prog. Neurobiol. 67, 203.
- Levi-Montalcini, R. (1966) Harvey Lect. 60, 217.
- Farinas, I. et al. (1998) Neuron 21, 325.
- Levi-Montalcini, R. et al. (1996) Trends Neurosci. 19, 514.
- Bradshaw, R.A. (1978) Ann. Rev. Biochem. 47, 191.
- Bocchini, V. and Angeletti, P.U. (1969) Proc. Natl. Acad. Sci. U.S.A. 64, 787.
- McDonald, N.Q. et al. (1991) Nature 354, 411.
- Huang, E.J. and Reichardt, L.F. (2001) Annu. Rev. Neurosci. 24, 677.
- Freund V. and Frossard, N. (2004) Prog. Brain Res. 146, 335.
- Raychaudhuri, S.P. and Raychaudhuri, S.K. (2004) Prog. Brain Res. 146, 433.
- Kawamoto, K. and Matsuda, H. (2004) Prog. Brain. Res. 146, 369.
- Teng, K.K. and Hempstead, B.L. (2004) Cell Mol. Life Sci. 61, 35.
The neurotrophins ("neuro" means nerve and "trophe" means nutrient) are a family of soluble, basic growth factors which regulate neuronal development, maintenance, survival and death in the CNS and the PNS.1 NGF, the first member of the family to be discovered, was originally purified as a factor able to support survival of sympathetic and sensory spinal neurons in culture.2 It is synthesized and secreted by sympathetic and sensory target organs and provides trophic support to neurons as they reach their final target.3 Neurotrophin secretion also increases in the nervous system following injury. Schwann cells, fibroblasts, and activated mast cells normally synthesize NGF constitutively, however direct trauma and induced cytokines combine to increase neurotrophin production in these cells after injury.4
NGF is purified in three forms: the 7S, 2.5S and β. The 7S, 130 kDa, form occurs naturally in mouse submaxillary glands, and is a multimeric protein composed of two α, one β and two γ subunits. The name is derived from its sedimentation co-efficient, 7S. The biologically active subunit is the β, which is a 26 kDa dimer composed of two identical 120 amino acid chains held together by hydrophobic interactions.5 The 2.5S form is 9 amino acids shorter than the β form, because of proteolysis that occurs during the purification process.6 The structural hallmark of all the neurotrophins is the characteristic arrangement of the disulfide bridges known as the cysteine knot, which has been found in other growth factors such as PDGF.7 There is a 95.8% homology between the rat and mouse forms, and a 85% homology between the human and mouse.
NGF has been shown to regulate neuronal survival, development function and plasticity.8 Recently, involvement of NGF in processes not involving neuronal cells has been shown, such as asthma,9 psoriasis10 and wound healing.11
The biological effects of NGF are mediated by two receptors:
TrkA, which is specific for NGF, and p75, which binds all the neurotrophins.12
Application key:
Species reactivity key:
Alomone Labs is pleased to offer a highly specific monoclonal antibody directed against an epitope of mouse NGF. Mouse NGF/proNGF Neutralizing Antibody (#ALM-006) can be used in indirect ELISA and neutralization assays to block the biological effects of both NGF and proNGF. It was designed to recognize NGF and proNGF from human, rat and mouse samples.
