Overview
- McDonough, S.I. et al. (1996) J. Neurosci. 16, 2612
- Herrero, C.J. et al. (1999) Br. J. Pharmacol. 127, 1375.
 Alomone Labs ω-Conotoxin MVIIC inhibits CaV2.2 heterologously expressed in Xenopus oocytes.A. Time course of ω-Conotoxin MVIIC (#C-150) blocking action on CaV2.2 channels maximum current (expressing α1B + α2δ1 + β1 subunits). Maximum current amplitudes were plotted as a function of time. Membrane potential was held at -100 mV and oocytes were stimulated by a 100 ms voltage ramp to 60 mV. 50 nM ω-Conotoxin MVIIC was perfused as indicated by the bar (green) for 160 sec. B. Superimposed examples of CaV2.2 channel peak current in the absence (control) and presence (green) of 50 nM ω-Conotoxin MVIIC (taken from experiment in A).
Alomone Labs ω-Conotoxin MVIIC inhibits CaV2.2 heterologously expressed in Xenopus oocytes.A. Time course of ω-Conotoxin MVIIC (#C-150) blocking action on CaV2.2 channels maximum current (expressing α1B + α2δ1 + β1 subunits). Maximum current amplitudes were plotted as a function of time. Membrane potential was held at -100 mV and oocytes were stimulated by a 100 ms voltage ramp to 60 mV. 50 nM ω-Conotoxin MVIIC was perfused as indicated by the bar (green) for 160 sec. B. Superimposed examples of CaV2.2 channel peak current in the absence (control) and presence (green) of 50 nM ω-Conotoxin MVIIC (taken from experiment in A).
- McDonough, S.I. et al. (1996) J. Neurosci. 16, 2612.
- Liu, H. et al. (1996) J. Biol. Chem. 23, 13804.
- Stocker, J.W. et al. (1997) J. Neurosci. 17, 3002.
- Herrero, C.J. et al. (1999) Br. J. Pharmacol. 127, 1375.
- Newcomb, R. and Palma, A. (1994) Brain. Res. 638, 95.
- Vega, T. et al. (1995) Eur. J. Pharmacol. 276, 231.
- Hirata, H. et al. (1997) Eur. J. Pharmacol. 321, 217.
ω-Conotoxin MVIIC blocks CaV2.1 (α1A, P/Q-type) and CaV2.2 (α1B, N-type) channels.1 The toxin binds with high affinity to CaV2.1 and with lower affinity to CaV2.2 in rabbit brain.2 However, the block by ω-Conotoxin-MVIIC of N-type channels in DRG neurons developed much faster than the block of P-type currents in Purkinje cells.1 The effect of the toxin is modulated by voltage (i.e. it is more potent for inactivated channels).3
In addition this toxin was reported to block nicotinic receptors (transiently expressed in Xenopus oocytes) with IC50 of 1.3 µM.4 It was also shown to inhibit K+-induced 3H-GABA release in hippocampus in vivo.5 This effect was with high affinity (50% block, 200 nM). The toxin was used to inhibit synaptic transmission in several peripheral preparations.6,7
ω-Conotoxin-MVIIC (#C-150) is a highly pure, synthetic, and biologically active peptide toxin.
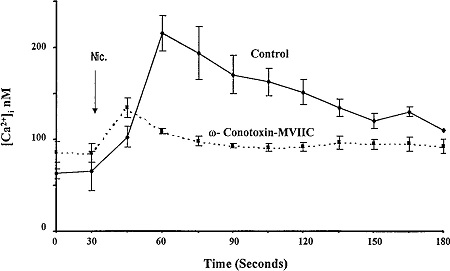
Alomone Labs ω-Conotoxin MVIIC partially affects [Ca2+]i in nicotine-treated PC12 cells.
Cells were loaded with fura 2 and incubated without (control) or with 500 nM ω-Conotoxin MVIIC (#C-150). Intracellular Ca2+ was measured at 15 sec intervals before and after addition of nicotine. ω-Conotoxin MVIIC reduced [Ca2+]i in response to nicotine by 65%.Adapted from Gueorguiev, V.D. et al. (1999) Am. J. Physiol. 276, C54. with permission of the American Physiological Society.
Applications
Citations
- Eltes, T. et al. (2017) J. Neurosci. 37, 1910.
- Pollak, J. et al. (2017) PLoS ONE 12, e0172884.
- Resch, J.M. et al. (2017) Neuron 96, 190.
- Forostyak, O. et al. (2016) Stem Cell Res. 16, 622.
- Evans, R.C. et al. (2015) J. Neurophysiol. 113, 2979.
- Gerencser, A.A. et al. (2015) Biochem. J. 471, 111.
- Perez-Burgos, A. et al. (2014) FASEB J. 28, 3064.
- Craviso, G.L. et al. (2010) Cell. Mol. Neurobiol. 30, 1259.
- Chu, J.Y. et al. (2009) Proc. Natl. Acad. Sci. U.S.A. 106, 15961.
- Chen, S. and Yaari, Y. (2008) J. Physiol. 586, 1351.
- Wu, W.W. et al. (2008) J. Neurophysiol. 100, 1897.
- Gueorguiev, V.D. et al. (1999) Am. J. Physiol. 276, C54.

