Overview
- Bocchini, V. and Angeletti, P.U. (1969) Proc. Natl. Acad. Sci. U.S.A. 64, 787.
- Huang, E.J. and Reichardt, L.F. (2001) Annu. Rev. Neurosci. 24, 677.
- Teng, K.K. and Hempstead, B.L. (2004) Cell Mol. Life Sci. 61, 35.
 TrkA in mouse cerebellar cortex.A. Fixed brain sections were incubated with mouse NGF 2.5S-Biotin (#N-240-B) followed by streptavidin DyLight-488 (green). Staining of Bergmann glial processes (vertical arrow) in the molecular layer (Mol) and of Purkinje cell soma (horizontal arrow). B. The same sections were incubated with Anti-TrkA (extracellular)-ATTO Fluor-633 Antibody (#ANT-018-FR). C. Merged image of A and B shows co-localization of NGF and TrkA mostly in the Purkinje layer (horizontal arrow). Nissl staining is used as the counterstain (blue).
TrkA in mouse cerebellar cortex.A. Fixed brain sections were incubated with mouse NGF 2.5S-Biotin (#N-240-B) followed by streptavidin DyLight-488 (green). Staining of Bergmann glial processes (vertical arrow) in the molecular layer (Mol) and of Purkinje cell soma (horizontal arrow). B. The same sections were incubated with Anti-TrkA (extracellular)-ATTO Fluor-633 Antibody (#ANT-018-FR). C. Merged image of A and B shows co-localization of NGF and TrkA mostly in the Purkinje layer (horizontal arrow). Nissl staining is used as the counterstain (blue).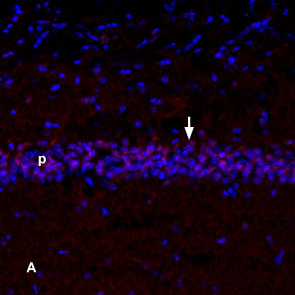 NGF localization in mouse hippocampus.Localization of NGF in mouse frozen brain floating sections using mouse NGF 2.5S-Biotin (#N-240-B), (10 nM), followed by Extravidin conjugated to Cy3 (red). Blue indicates DAPI nissl counterstain. Mouse NGF labeling (red) is restricted to the pyramidal layer (arrow). P = Purkinje layer.
NGF localization in mouse hippocampus.Localization of NGF in mouse frozen brain floating sections using mouse NGF 2.5S-Biotin (#N-240-B), (10 nM), followed by Extravidin conjugated to Cy3 (red). Blue indicates DAPI nissl counterstain. Mouse NGF labeling (red) is restricted to the pyramidal layer (arrow). P = Purkinje layer.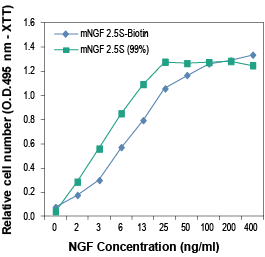 Alomone Labs mouse NGF 2.5S-Biotin promotes survival in PC12 cells.Cells were grown in the absence of serum and in the presence of varying concentrations of Native mouse NGF 2.5S protein (99%) (#N-240) (green) or mouse NGF 2.5S-Biotin (#N-240-B) (blue). After 24 h cell survival was measured by XTT method and calculated as a relative percentage of the control and plotted against concentrations used.
Alomone Labs mouse NGF 2.5S-Biotin promotes survival in PC12 cells.Cells were grown in the absence of serum and in the presence of varying concentrations of Native mouse NGF 2.5S protein (99%) (#N-240) (green) or mouse NGF 2.5S-Biotin (#N-240-B) (blue). After 24 h cell survival was measured by XTT method and calculated as a relative percentage of the control and plotted against concentrations used. Alomone Labs mouse NGF 2.5S-Biotin promotes dose-dependent and specific binding of Avidin-HRP in PC12 cells but not in HEK293 cells.Cells in suspension were incubated with different concentrations of mouse NGF 2.5S-Biotin (#N-240-B) for 15 min at 4°C, followed by Avidin-HRP (1:500, 1 h, 4°C). Binding of Avidin-HRP to Biotin was determined by measuring the absorbance at 450 nm which was plotted against mouse NGF 2.5S-Biotin concentration for both cell lines (error bars indicating standard deviation, n = 3).
Alomone Labs mouse NGF 2.5S-Biotin promotes dose-dependent and specific binding of Avidin-HRP in PC12 cells but not in HEK293 cells.Cells in suspension were incubated with different concentrations of mouse NGF 2.5S-Biotin (#N-240-B) for 15 min at 4°C, followed by Avidin-HRP (1:500, 1 h, 4°C). Binding of Avidin-HRP to Biotin was determined by measuring the absorbance at 450 nm which was plotted against mouse NGF 2.5S-Biotin concentration for both cell lines (error bars indicating standard deviation, n = 3).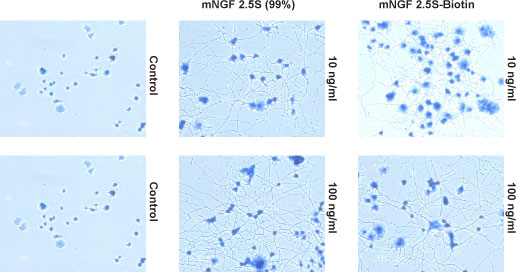 Alomone Labs mouse NGF 2.5S-Biotin promotes neurite outgrowth.Cells were grown on collagen-coated plates in the absence (Control) or presence of 10 ng/ml or 100 ng/ml Native mouse NGF 2.5S protein (99%) (#N-240) or mouse NGF 2.5S-Biotin (#N-240-B). The development of neurites over a period of 6 days was visualized by Methylene Blue staining.
Alomone Labs mouse NGF 2.5S-Biotin promotes neurite outgrowth.Cells were grown on collagen-coated plates in the absence (Control) or presence of 10 ng/ml or 100 ng/ml Native mouse NGF 2.5S protein (99%) (#N-240) or mouse NGF 2.5S-Biotin (#N-240-B). The development of neurites over a period of 6 days was visualized by Methylene Blue staining.
- Roux, P.P. and Barker P.A. (2002) Prog. Neurobiol. 67, 203.
- Levi-Montalcini, R. (1966) Harvey Lect. 60, 217.
- Kalb, R. (2005) Trends Neurosci. 28, 5.
- Huang, E.J. and Reichardt, L.F. (2001) Annu. Rev. Neurosci. 24, 677.
- Farinas, I. et al. (1998) Neuron 21, 325.
- Levi-Montalcini, R. et al. (1996) Trends Neurosci. 19, 514.
- McDonald, N.Q. et al. (1991) Nature 354, 411.
- Freund V. and Frossard, N. (1994) Prog. Brain Res. 146, 335.
- Raychaudhuri, S.P. and Raychaudhuri, S.K. (2004) Prog. Brain Res. 146, 433.
- Kawamoto, K. and Matsuda, H. (2004) Prog. Brain Res. 146, 369.
- Rosenberg, M.B. et al. (1986) J. Neurochem. 46, 641.
- Rosenberg, M.B. et al. (1987) J. Neurochem. 48, 865.
- Bronfman, F.C. et al. (2003) J. Neurosci. 23, 3209.
- George, P.M. et al. (2006) Adv. Materials 18, 577.
- Cui, B. et al. (2007) Proc. Natl. Acad. Sci. U.S.A. 104, 13666.
- Perlson, E. et al. (2009) J. Neurosci. 29, 9903.
- Teng, K.K. and Hempstead, B.L. (2004) Cell Mol. Life Sci. 61, 35.
The neurotrophins ("neuro" means nerve and "trophe" means nutrient) are a family of soluble, basic protein growth factors which regulate neuronal development, maintenance, survival and death in the CNS and PNS1.
NGF, the first member of the family to be discovered, was originally purified as a factor supporting and regulating survival, development, function and plasticity of sympathetic and sensory spinal neurons in central and peripheral nervous systems in vivo as well as in vitro2-4. It is synthesized and secreted by sympathetic and sensory target organs and provides trophic support to neurons as they reach their final target5.
Neurotrophin secretion increases in the nervous system following injury. Schwann cells, fibroblasts, and activated mast cells normally synthesize NGF constitutively, however, direct trauma and induction of cytokines combine to increase neurotrophin production in these cells after injury6.
NGF is purified in three forms: the 7S, 2.5S and β, in which the biologically active subunit is the β subunit. The structural hallmark of all the neurotrophins is the characteristic arrangement of the disulfide bridges known as the cysteine knot, which has been found in other growth factors such as Platelet-derived growth factor7.
Additionally, the involvement of NGF was recently discovered in processes such as asthma8, psoriasis9 and wound healing10. The biological effects of NGF are mediated by two receptors: TrkA, which is specific for NGF, and p75NTR, which binds all the neurotrophins11.
For the past three decades, biotinylated derivatives of NGF have been widely used in the literature as useful probes for the study of the binding and the initial intracellular processing of this growth factor by cells bearing TrkA NGF receptor12. First studies using 125I-NGF have visualized the intracellular location of NGF12. A biotinylated derivative of NGF that retains biological activity, in conjunction with the appropriate avidin conjugates and fluorescent or confocal microscopic techniques, improve the sensitivity and resolution in many applications. Some examples of applications are: targeting of liposomes containing biotinylated NGF to cells bearing TrkA NGF receptors13; elucidating the kinetics and route of ligand-induced internalization of the p75 receptor through signaling endosomes in cycling and differentiated PC12 cells using biotinylated derivative of NGF14; electrically controlled NGF-Biotin delivery from biotin-doped conductive polymer15; studying the retrograde axonal transport mechanism of NGF signals from the axon terminal to the cell body16,17.
mouse NGF 2.5S-Biotin (#N-240-B) is a highly pure and biologically active conjugated protein.
✓ Targeting liposomes containing biotinylated NGF to cells bearing TrkA receptors
✓ Elucidating the kinetics and route of ligand-induced internalization of the p75 receptor
✓ Studying the retrograde axonal transport mechanism of NGF signals from the axon terminal to the cell body
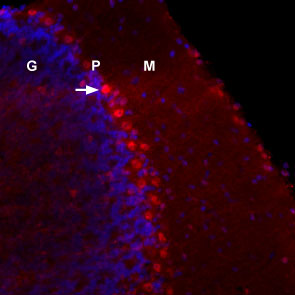
NGF localization in mouse cerebellumLocalization of NGF in mouse frozen brain floating sections using mouse NGF 2.5S-Biotin (#N-240-B), (10 nM), followed by Extravidin conjugated to Cy3 (red). Blue indicates DAPI nissl counterstain. Mouse NGF labeling (red) is restricted to Purkinje cells (arrow). G = Granule layer, P = Purkinje layer, M = Molecular layer.
Retrograde transport of Alomone Labs mouse NGF 2.5S-Biotin conjugated to Qdot in DRG explants.
Dorsal root ganglia explant from E13.5 mice were grown 3 days in compartmental chambers. 4 nM of mouse NGF 2.5S-Biotin (#N-240-B) conjugated to Qdot605 were added to the distal axon chamber. Retrograde transport of Qdot-NGF was imaged along one axon. Pictures were taken every 2 sec at x60 magnification.
Applications
Citations
- KC, R. et al. (2016) Ann. Rheum. Dis. In press.
- Tanaka, Y. et al. (2016) Neuron 90, 1215.
- Villarin, J.M. et al. (2016) Nat. Commun. 7, 13865.
