Overview
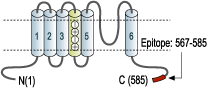
- Peptide CKESPVIAKYMPTEAVRVT, corresponding to amino acid residues 567-585 of rat KV3.1b (Accession P25122). Intracellular, C-terminus.
 Western blot analysis of rat brain membranes:1. Anti-KV3.1b (KCNC1) Antibody (#APC-014), (1:200).
Western blot analysis of rat brain membranes:1. Anti-KV3.1b (KCNC1) Antibody (#APC-014), (1:200).
2. Anti-KV3.1b (KCNC1) Antibody, preincubated with Kv3.1b/KCNC1 Blocking Peptide (#BLP-PC014).
- Transfected COS-7 cells (Wu, H. et al. (1998) J. Cell Sci. 111, 2365.).
- Rat brain sections. Mouse medial nucleus of the trapezoid body (MNTB) (1:500) (Leao, K.E. et al. (2010) Eur. J. Neurosci. 32, 1658.).
- Rat neural progenitor cells (NPCs) (1:200) (Liebau, S. et al. (2006) J. Neurochem. 99, 426.).
- Rettig, J. et al. (1992) EMBO J. 11, 2473
- Espinosa, F. et al. (2001) J. Neurosci. 21, 6657.
- Rudy, B. et al. (2001) Trends Neurosci. 24, 517.
KV3.1 is a member of the voltage-gated K+ channel superfamily. Together with the related proteins KV3.2, KV3.3 and KV3.4 they constitute the Shaw type subfamily family.1
As with all KV channels, KV3.1 possesses the signature structure of the voltage-dependent K+ channels: six membrane-spanning domains with intracellular N and C termini. The functional Kv channel is a tetramer that can either be a homomer or a heteromer of KV3 subunits.
KV3 subfamily members inactivate very rapidly and therefore are thought to play a role in the repolarization of action potentials and to facilitate repetitive high frequency firing.3
KV3.1 is highly expressed in the brain but has been also detected in peripheral organs such as lung skeletal muscle and testis.
KV3.1 and KV3.2 are highly enriched in neurons that fire at high frequencies, such as fast spiking interneurons of the cortex and hippocampus and neurons in the globus pallidus. Their unusually rapid activation and deactivation rates allow channels containing KV3.1 and KV3.2 subunits to repolarize action potentials quickly thus minimizing the rate of recovery of sodium channel inactivation.3
Lack of KV3.1 channel subunits is mainly responsible for constitutively increased locomotor activity and sleep loss.2
Application key:
Species reactivity key:
Anti-KV3.1b (KCNC1) Antibody (#APC-014) is a highly specific antibody directed against an epitope of the rat protein. The antibody can be used in western blot, immunohistochemistry, immunocytochemistry, and immunoprecipitation applications. It has been designed to recognize KV3.1b from rat, mouse, and human samples.
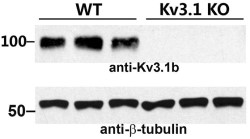
Knockout validation of Anti-KV3.1b (KCNC1) Antibody in mouse brain.Western blot analysis of mouse brain lysates using Anti-KV3.1b (KCNC1) Antibody (#APC-014). KV3.1b immunodetection is abolished in KV3.1-/- mice. β-tubulin is used as a loading control.Adapted from Jukkola, P. et al. (2017) Front. Mol. Neurosci. 10, 344.
Applications
Citations
 Co-localization of KV3.1b and KIF5 in rat hippocampal neuronsImmunocytochemical staining of rat hippocampal neurons with Anti-KV3.1b (KCNC1) Antibody (APC-014) and anti-KIF5 antibody. Arrows in right panel depict colocalization between the two proteins.
Co-localization of KV3.1b and KIF5 in rat hippocampal neuronsImmunocytochemical staining of rat hippocampal neurons with Anti-KV3.1b (KCNC1) Antibody (APC-014) and anti-KIF5 antibody. Arrows in right panel depict colocalization between the two proteins.
Adapted from Barry, J. et al. (2013) with permission of The Company of Biologists LTD.
- Western blot analysis of mouse brain lysates. Also tested in KV3.1-/- mice.
Jukkola, P. et al. (2017) Front. Mol. Neurosci. 10, 344. - Western blot analysis of mouse brain lysates. Immunohistochemical staining of mouse coronal cerebellum sections. Immunocytochemical staining of mouse cultured hippocampal and cerebellar neurons. Tested in KV3.1b knockout mice and KV3.1b knockout mice cultured cerebellar neurons.
Barry, J. et al. (2013) J. Cell Sci. 126, 2027.
- Mouse brain lysates. Also tested in KV3.1-/- mice.
Jukkola, P. et al. (2017) Front. Mol. Neurosci. 10, 344. - Rat lung lysate.
Lv, Y. et al. (2013) Am. J. Physiol. 305, L856. - Mouse brain lysates. Also tested in KV3.1b knockout mice.
Barry, J. et al. (2013) J. Cell Sci. 126, 2027.
- Transfected COS-7 cells.
Wu, H. et al. (1998) J. Cell Sci. 111, 2365.
- Rat lumbar spinal cord sections.
Wolff, M. et al. (2016) Neurosci. Res. 109, 16. - Mouse brain sections (1:800).
Blosa, M. et al. (2015) J. Physiol. 593, 4341. - Mouse coronal cerebellum sections. Also tested in same sections from KV3.1b knockout mice.
Barry, J. et al. (2013) J. Cell Sci. 126, 2027. - Mouse medial nucleus of the trapezoid body (MNTB) (1:500).
Leao, K.E. et al. (2010) Eur. J. Neurosci. 32, 1658.
- Eltes, T. et al. (2017) J. Neurosci. 37, 1910.
- Brooks, N. L. et al. (2006) FEBS J. 273, 3287.
- Leao, R.N. et al. (2006) J. Physiol. 571, 563.
- Tiwari-Woodruff. S. et al. (2006) Am. J. Physiol. Cell Physiol. 291, 687.
- Dallas, M.L. et al. (2005) J. Physiol. 562.3, 655.
- Devaux, J.J and Scherer S.S. (2005) J. Neurosci. 25, 1470.
- Itri, J.N. et al. (2005) Nature Neurosci. 8, 650.
- Wang, J. et al. (2005) Am. J. Physiol. Lung Mol. Cell Physiol. 288, L1049.
- Brooke, R.E. et al. (2004) Eur. J. Neurosci. 20, 3313.
- Ruttiger, L. et al. (2004) PNAS 101, 12922.
- Shevchenko, T. et al. (2004) J. Neurophysiol. 92, 3043.
- Baranauskas, G. et al. (2003) Nature Neurosci. 6, 258.
- Devaux, J. et al. (2003) J. Neurosci. 23, 4509.
- Dodson, P.D. et al. (2003) J. Physiol. 550.1 27.
- Adamson, C.L. et al (2002) J. Neuroscience 22, 1385.
- Felix, R. et al. (2002) Zygote 10, 183.
- Moss, F.J. et al. (2002) EMBO J. 21, 1514.
- Osipenko, O.N. et al. (2000) Circ Res. 86, 534.
- Yue, L. et al. (2000) J. Physiol. 527.3, 467.
- Gelband, C.H. et al. (1999) Circ Res. 84, 352.
- Hartig, W. et al. (1999) Brain Res. 842, 15.
- Sobko, A. et al. (1998) J. Neurosci.18, 10398.
- Sobko, A. et al. (1998) EMBO J. 17, 4723.
