Overview
- Bourinet, E. et al. (2001) Biophys. J. 81, 79.
- Kimm, T. and Bean, B.P. (2014) J. Neurosci. 34, 9182.
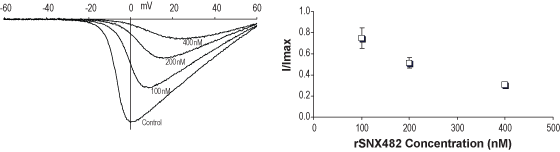 Alomone Labs SNX-482 inhibits CaV2.3 channels heterologously expressed in Xenopus oocytes.CaV2.3 channel subunits co-expressed in Xenopus oocytes. Using TEVC, membrane potential was held at -100 mV. Ba2+ (10 mM) currents via CaV2.3 channels were elicited by 40 ms long voltage ramps from -100 to +60 mV, delivered every 10 seconds. Left: Representative current traces before and following the application of 100, 200 and 400 nM SNX-482 (#RTS-500), as indicated. Right: Dose-response for SNX-482 (n = 2-6).
Alomone Labs SNX-482 inhibits CaV2.3 channels heterologously expressed in Xenopus oocytes.CaV2.3 channel subunits co-expressed in Xenopus oocytes. Using TEVC, membrane potential was held at -100 mV. Ba2+ (10 mM) currents via CaV2.3 channels were elicited by 40 ms long voltage ramps from -100 to +60 mV, delivered every 10 seconds. Left: Representative current traces before and following the application of 100, 200 and 400 nM SNX-482 (#RTS-500), as indicated. Right: Dose-response for SNX-482 (n = 2-6).
- Newcomb, R. et. al. (1998) Biochemistry 37, 15353.
- Bourinet, E. et al. (2001) Biophys. J. 81, 79.
- Wang, G. et al. (1999) J. Neurosci. 19, 9235.
- Arroyo, G. et al. (2003) Eur. J. Pharmacol. 475, 11.
- Kimm, T. and Bean, B.P. (2014) J. Neurosci. 34, 9182.
SNX-482 is a peptidyl toxin originally isolated from venom of the spider Hysterocrates gigas. Native SNX-482 blocks specifically CaV2.3 (α1E, R-type) channels1 in a voltage-dependent manner. The block is reversible only upon application of strong voltage to facilitate unbinding.2 SNX-482 inhibits human CaV2.3 channels stably expressed in a mammalian cell line. An IC50 of 15-30 nM was obtained for block of CaV2.3 channel, using either patch clamp electrophysiology or K+-evoked Ca2+ flux. At low nanomolar concentrations, SNX-482 also blocked a native R-type Ca2+ current in rat neurohypophyseal nerve terminals, but concentrations of 200-500 nM had no effect on R-type Ca2+ currents in several types of rat central neurons.1 SNX-482 was also used to demonstrate the contribution of CaV2.3 channels to transmitter release.3 Recently it was shown that higher concentrations of SNX-482 also block CaV2.1 channels in chromaffin cells.4
SNX-482 was found to be the most potent blocker to date for KV4.3 channel with IC50 < 3 nM5.
SNX-482 (#RTS-500) is a highly pure, recombinant, and biologically active peptide toxin.
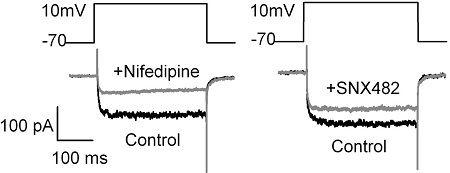
Alomone Labs Nifedipine and SNX-482 block L-type and R-type CaV channels respectively in rat pancreatic INS-1 832/13 β-cells.Representative CaV currents from INS-1 832/13 cells before and after treatment with Nifedipine (#N-120), a general L-type CaV channel blocker and SNX-482 (#RTS-500), a CaV2.3 channel blocker. 52% of the currents were blocked by Nifedipine (n = 8; p < 0.05) while 31% (n = 10; p < 0.001) of currents were blocked by SNX-482. Cells were held at −70 mV for 2 min after formation of whole-cell mode, and currents elicited by stepped 300 or 500 millisecond depolarizations in 10 mV increments.Adapted from Xie, L. et al. (2016) PLoS ONE 11, e0147862. with permission of PLoS.
Applications
Citations
 SNX-482 blocks KV4.3 currents heterologously expressed in HEK 293 cells.Effect of 3 nM (top) and 60 nM (bottom) SNX-482 (#RTS-500) on current carried by cloned KV4.3 channels expressed in HEK-293 cells (left panels). Effect of 3 nM (top) and 60 nM (bottom) SNX-482 on current carried by cloned CaV2.3 channels expressed in HEK-293 cells. Recordings at 23°C.
SNX-482 blocks KV4.3 currents heterologously expressed in HEK 293 cells.Effect of 3 nM (top) and 60 nM (bottom) SNX-482 (#RTS-500) on current carried by cloned KV4.3 channels expressed in HEK-293 cells (left panels). Effect of 3 nM (top) and 60 nM (bottom) SNX-482 on current carried by cloned CaV2.3 channels expressed in HEK-293 cells. Recordings at 23°C.
Adapted from Kimm, T. and Bean, B.P. (2014) with permission of the Society for Neuroscience.
- Rat INS-1 832/12 cells (whole-cell calcium currents).
Luan, C. et al. (2019) Commun. Biol. 2, 106. - Rat DRGs (patch clamp).
Moutal, A. et al. (2018) Neuroscience 381, 79.
- Resch, J.M. et al. (2017) Neuron 96, 190.
- Sugino, S. et al. (2016) J. Neurophysiol. 115, 1577.
- Xie, L. et al. (2016) PLoS ONE 11, e0147862.
- Gerencser, A.A. et al. (2015) Biochem. J. 471, 111.
- Rudolph, S. et al. (2015) J. Neurosci. 35, 15492.
- He, S. et al. (2014) J. Neurosci. 34, 5261.
- Kimm, T. and Bean, B.P. (2014) J. Neurosci. 34, 9182.
- Lv, P. et al. (2014) J. Neurosci. 34, 7383.
- Jones, S.L. and Stuart, G.J. (2013) J. Neurosci. 33, 19396.
- Abitbol, K. et al. (2012) J. Physiol. 590.13, 2977.
- Anderson, T.M. et al. (2012) J. Neurophysiol. 107, 103.
- Lv, P. et al. (2012) J. Neurosci. 32, 163143.
- Tzour, A. et al. (2012) J. Neuroendocrinol. 25, 76.
- Chalifoux, J.R. and Carter, A.G. (2011) J. Neurosci. 31, 16435.
- Hao, M.M. et al. (2011) J. Neurosci. 31, 15352.
- Myoga, M.H. and Regehr, W.G. (2011) J. Neurosci. 31, 5235.
- Castro, A. et al. (2009) J. Comp. Neurol. 513, 188.
- Chu, J.Y. et al. (2009) Proc. Natl. Acad. Sci. U.S.A. 106, 15961.
- Yang, L. and Stephens, G.J. (2009) Cell Calcium 46, 248.
- Braun, M. et al. (2008) Diabetes 57, 1618.

