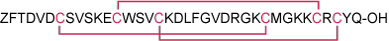Overview
Cat #:
STI-400
Alternative Name K+ channel toxin α-KTx 1.3, IbTx
Lyophilized Powder yes
Origin Synthetic peptide
MW: 4230.9 Da
Purity: >98% (HPLC)
Effective concentration 50-100 nM.
Sequence ZFTDVDCSVSKECWSVCKDLFGVDRGKCMGKKCRCYQ.
Modifications Disulfide bonds between: Cys7-Cys28, Cys13-Cys33, and Cys17-Cys35. Z = Pyrrolidone carboxylic acid.
Molecular formula C179H274N50O55S7
CAS No.: 129203-60-7
Activity Iberiotoxin is a potent selective blocker of the high conductance Ca2+-activated K+ channels (maxi-K).
Shipping and storage Shipped at room temperature. Product as supplied can be stored intact at room temperature for several weeks. For longer periods, it should be stored at -20°C.
Solubility Any aqueous buffer. Centrifuge all product preparations before use (10000 x g 5 min).
Storage of solutions Up to two weeks at 4°C or three months at -20°C.
Our bioassay
 Alomone Labs Iberiotoxin inhibits KCa1.1 channels (mSlo) heterologously expressed in Xenopus oocytes.A. Time course of KCa1.1 channel current amplitude before (black), during (green, marked by horizontal bar) application of 100 nM Iberiotoxin (#STI-400) for 200 sec and upon wash of the toxin. Currents were elicited every 10 sec by ramp stimulation to +100 mV from a holding of –100 mV for 100 msec. B. Superimposed example current responses before (black) and during (green) application of 100 nM Iberiotoxin (taken from the experiment in A).
Alomone Labs Iberiotoxin inhibits KCa1.1 channels (mSlo) heterologously expressed in Xenopus oocytes.A. Time course of KCa1.1 channel current amplitude before (black), during (green, marked by horizontal bar) application of 100 nM Iberiotoxin (#STI-400) for 200 sec and upon wash of the toxin. Currents were elicited every 10 sec by ramp stimulation to +100 mV from a holding of –100 mV for 100 msec. B. Superimposed example current responses before (black) and during (green) application of 100 nM Iberiotoxin (taken from the experiment in A).
References - Scientific background
- Galvez, A. et al. (1990) J. Biol. Chem. 265, 11083.
- Koschak, A. et al. (1997) Biochemistry 36, 1943.
- Meera, P. et al. (2000) Proc. Natl. Acad. Sci. U.S.A. 97, 5562.
Scientific background Iberiotoxin is a 37 amino acid peptidyl toxin isolated from the scorpion Mesobuthus tamulus and was shown to block large conductance Ca2+-activated K+ channels in smooth muscle cells1. Later it was shown to specifically block KCa1.1 (Slo) channels with Ki of about 1 nM2. In addition, experiments with cloned KCa1.1 channels demonstrate the strong effect of the sloβ subunits on the potency of block by Iberiotoxin3.
Target KCa1.1 K+ channels
Peptide Content: 100%
Lyophilized Powder
Iberiotoxin (#STI-400) is a highly pure, synthetic, and biologically active peptide toxin.
Image & Title 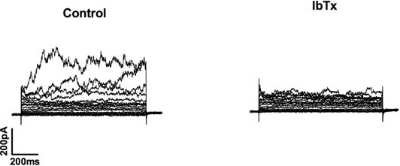
Alomone Labs Iberiotoxin blocks BKCa channels in human chorionic plate arterial smooth muscle cells.Inhibition of BKCa channels with Iberiotoxin (#STI-400) which abolished outward currents at +80 mV by 61 ± 12%. Cells were voltage clamped at -60 mV and step depolarised from -70 mV to +80 mV for 500 ms in 10 mV increments and repolarised to -40 mV.Adapted from Brereton, M.F. et al. (2013) PLoS ONE 8, e57451. with permission of PLoS.

Alomone Labs Iberiotoxin blocks BKCa channels in human chorionic plate arterial smooth muscle cells.Inhibition of BKCa channels with Iberiotoxin (#STI-400) which abolished outward currents at +80 mV by 61 ± 12%. Cells were voltage clamped at -60 mV and step depolarised from -70 mV to +80 mV for 500 ms in 10 mV increments and repolarised to -40 mV.Adapted from Brereton, M.F. et al. (2013) PLoS ONE 8, e57451. with permission of PLoS.
For research purposes only, not for human use
Last Update: 21/05/2024
Applications
Citations
Citations
Published figures using this product
 Blocking BK channels mimics the mAChR activation effects.A. and B. uEPSC (left), uEPSP (middle), and ΔGuEPSP/Gsat (right) measured in the presence of Apamin (#STA-200) (A) and Iberiotoxin (#STI-400) (B). The range of amplitudes ±SEM of the uEPSP and ΔGuEPSP/Gsat measured in control conditions are shown by the gray shaded bars. C. Summary of the uEPSC amplitude, uEPSP amplitude, uEPSP decay time constant, and ΔGuEPSP/Gsat in each pharmacological condition.
Blocking BK channels mimics the mAChR activation effects.A. and B. uEPSC (left), uEPSP (middle), and ΔGuEPSP/Gsat (right) measured in the presence of Apamin (#STA-200) (A) and Iberiotoxin (#STI-400) (B). The range of amplitudes ±SEM of the uEPSP and ΔGuEPSP/Gsat measured in control conditions are shown by the gray shaded bars. C. Summary of the uEPSC amplitude, uEPSP amplitude, uEPSP decay time constant, and ΔGuEPSP/Gsat in each pharmacological condition.
Adapted from He, S. et al. (2014) with permission of the Society for Neuroscience.
Electrophysiology
- Mouse brain slices (100 nM).
Lahiri, A. and Bevan, M.D. (2020) Neuron 106, 1. - Mouse α-cells (single cell).
Dickerson, M.T. et al. (2019) Am. J. Physiol. 316, E646. - Human beta cells.
Vierra, N.C. et al. (2017) Sci. Signal. 10, eaan2883.
Product citations
- Lu, M. et al. (2018) Am. J. Physiol. 314, C643.
- Hsu, H.T. et al. (2017) BMC Neurosci. 18, 6.
- Nelson, A.B. et al. (2017) Neuron 93, 211.
- Rosa, P. et al. (2017) J. Cell. Physiol. 232, 2478.
- Wolkart, G. et al. (2017) Br. J. Pharmacol. 174, 3640.
- Chen, M. et al. (2016) Physiol. Rep. 4, e12682.
- Li, Y. et al. (2016) PLoS ONE 11, e0155006.
- Margas, W. et al. (2016) Phil. Trans. R. Soc. B 371, 20150430.
- Rabbitt, R.D. et al. (2016) J. Neurophysiol. 116, 825.
- Webb, T.N. et al. (2016) Am. J. Physiol. 310, F15.
- Casale, A.E. et al. (2015) J. Neurosci. 35, 15555.
- Dell’Orco, J.M. et al. (2015) J. Neurosci. 35, 11292.
- Almog, M. and Korngreen, A. (2014) J. Neurosci. 34, 182.
- He, S. et al. (2014) J. Neurosci. 34, 5261.
- Lopez-Gonzalez, I. et al. (2014) Mol. Hum. Reprod. 20, 619.
- Rowan, M.J. et al. (2014) J. Neurosci. 34, 6611.
- Brereton, M.F. et al. (2013) PLoS ONE 8, e57451.
- Chen, M. et al. (2013) Mol. Neurobiol. 48, 794.
- Gonzalez Corrochano, R. et al. (2013) Br. J. Pharmacol. 169, 449.
- Qian, Z. et al. (2013) CNS Neurosci. Ther. 19, 954.
- Tsai, K.L. et al. (2013) Cell. Physiol. Biochem. 31, 938.
- Weisbrod, D. et al. (2013) Proc. Natl. Acad. Sci. U.S.A. 110, E1685.
- Zhong, L.R. et al. (2013) PLoS ONE 8, e78727.
- Hristov, K.L. et al. (2012) Am. J. Physiol. 302, C1632.
- So, E.C. et al. (2012) Eur. J. Pharmacol. 683, 1.
- Alle, H. et al. (2011) J. Neurosci. 31, 8001.
- Wu, S.N. et al. (2011) Cell. Physiol. Biochem. 28, 959.
- Kita, M. et al. (2010) Am. J. Physiol. 298, R1310.
- Liu, Y.C. et al. (2008) Eur. J. Pharmacol. 590, 93.
- Pelucchi, B. et al. (2008) J. Neurosci. Res. 86, 194.
- Begg, M. et al. (2001) J. Physiol. 531, 95.